Tuberculosis
| Tuberculosis | |
|---|---|
| Synonyms | phthisis, phthisis pulmonalis, consumption |
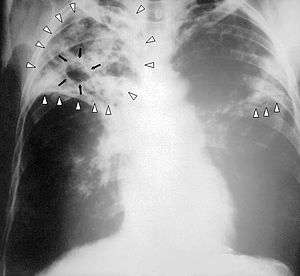 | |
| Chest X-ray of a person with advanced tuberculosis: Infection in both lungs is marked by white arrow-heads, and the formation of a cavity is marked by black arrows. | |
| Classification and external resources | |
| Specialty | Infectious disease, pulmonology |
| ICD-10 | A15–A19 |
| ICD-9-CM | 010–018 |
| OMIM | 607948 |
| DiseasesDB | 8515 |
| MedlinePlus | 000077 000624 |
| eMedicine | med/2324 emerg/618 radio/411 |
| Patient UK | Tuberculosis |
| MeSH | D014376 |
| Orphanet | 3389 |
Tuberculosis (TB) is an infectious disease caused by the bacterium Mycobacterium tuberculosis (MTB).[1] Tuberculosis generally affects the lungs, but can also affect other parts of the body. Most infections do not have symptoms, known as latent tuberculosis. About 10% of latent infections progress to active disease which, if left untreated, kills about half of those infected. The classic symptoms of active TB are a chronic cough with blood-containing sputum, fever, night sweats, and weight loss.[1] The historical term "consumption" came about due to the weight loss.[2] Infection of other organs can cause a wide range of symptoms.[3]
Tuberculosis is spread through the air when people who have active TB in their lungs cough, spit, speak, or sneeze.[1][4] People with latent TB do not spread the disease. Active infection occurs more often in people with HIV/AIDS and in those who smoke.[1] Diagnosis of active TB is based on chest X-rays, as well as microscopic examination and culture of body fluids. Diagnosis of latent TB relies on the tuberculin skin test (TST) or blood tests.[5]
Prevention of TB involves screening those at high risk, early detection and treatment of cases, and vaccination with the bacillus Calmette-Guérin vaccine.[6][7][8] Those at high risk include household, workplace, and social contacts of people with active TB.[8] Treatment requires the use of multiple antibiotics over a long period of time.[1] Antibiotic resistance is a growing problem with increasing rates of multiple drug-resistant tuberculosis (MDR-TB).[1]
One-third of the world's population is thought to be infected with TB.[1] New infections occur in about 1% of the population each year.[9] In 2014, there were 9.6 million cases of active TB which resulted in 1.5 million deaths. More than 95% of deaths occurred in developing countries. The number of new cases each year has decreased since 2000.[1] About 80% of people in many Asian and African countries test positive while 5–10% of people in the United States population tests positive by the tuberculin test.[10] Tuberculosis has been present in humans since ancient times.[11]
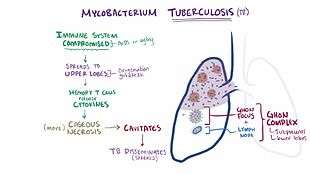
Signs and symptoms
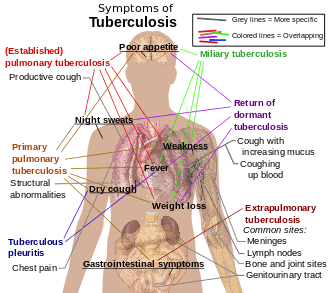
Tuberculosis may infect any part of the body, but most commonly occurs in the lungs (known as pulmonary tuberculosis).[3] Extrapulmonary TB occurs when tuberculosis develops outside of the lungs, although extrapulmonary TB may coexist with pulmonary TB.[3]
General signs and symptoms include fever, chills, night sweats, loss of appetite, weight loss, and fatigue.[3] Significant nail clubbing may also occur.[13]
Pulmonary
If a tuberculosis infection does become active, it most commonly involves the lungs (in about 90% of cases).[11][14] Symptoms may include chest pain and a prolonged cough producing sputum. About 25% of people may not have any symptoms (i.e. they remain "asymptomatic").[11] Occasionally, people may cough up blood in small amounts, and in very rare cases, the infection may erode into the pulmonary artery or a Rasmussen's aneurysm, resulting in massive bleeding.[3][15] Tuberculosis may become a chronic illness and cause extensive scarring in the upper lobes of the lungs. The upper lung lobes are more frequently affected by tuberculosis than the lower ones.[3] The reason for this difference is not clear.[10] It may be due to either better air flow,[10] or poor lymph drainage within the upper lungs.[3]
Extrapulmonary
In 15–20% of active cases, the infection spreads outside the lungs, causing other kinds of TB.[16] These are collectively denoted as "extrapulmonary tuberculosis".[17] Extrapulmonary TB occurs more commonly in immunosuppressed persons and young children. In those with HIV, this occurs in more than 50% of cases.[17] Notable extrapulmonary infection sites include the pleura (in tuberculous pleurisy), the central nervous system (in tuberculous meningitis), the lymphatic system (in scrofula of the neck), the genitourinary system (in urogenital tuberculosis), and the bones and joints (in Pott disease of the spine), among others. When it spreads to the bones, it is also known as "osseous tuberculosis",[18] a form of osteomyelitis.[10] Sometimes, bursting of a tubercular abscess through skin results in tuberculous ulcer.[19] An ulcer originating from nearby infected lymph nodes is painless, slowly enlarging and has an appearance of "wash leather".[20] A potentially more serious, widespread form of TB is called "disseminated tuberculosis", also known as miliary tuberculosis.[3] Miliary TB makes up about 10% of extrapulmonary cases.[21]
Causes
Mycobacteria
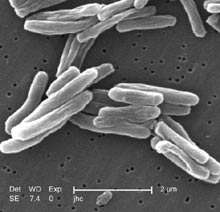
The main cause of TB is Mycobacterium tuberculosis, a small, aerobic, nonmotile bacillus.[3] The high lipid content of this pathogen accounts for many of its unique clinical characteristics.[22] It divides every 16 to 20 hours, which is an extremely slow rate compared with other bacteria, which usually divide in less than an hour.[23] Mycobacteria have an outer membrane lipid bilayer.[24] If a Gram stain is performed, MTB either stains very weakly "Gram-positive" or does not retain dye as a result of the high lipid and mycolic acid content of its cell wall.[25] MTB can withstand weak disinfectants and survive in a dry state for weeks. In nature, the bacterium can grow only within the cells of a host organism, but M. tuberculosis can be cultured in the laboratory.[26]
Using histological stains on expectorated samples from phlegm (also called "sputum"), scientists can identify MTB under a microscope. Since MTB retains certain stains even after being treated with acidic solution, it is classified as an acid-fast bacillus.[10][25] The most common acid-fast staining techniques are the Ziehl–Neelsen stain[27] and the Kinyoun stain, which dye acid-fast bacilli a bright red that stands out against a blue background.[28] Auramine-rhodamine staining[29] and fluorescence microscopy[30] are also used.
The M. tuberculosis complex (MTBC) includes four other TB-causing mycobacteria: M. bovis, M. africanum, M. canetti, and M. microti.[31] M. africanum is not widespread, but it is a significant cause of tuberculosis in parts of Africa.[32][33] M. bovis was once a common cause of tuberculosis, but the introduction of pasteurized milk has almost completely eliminated this as a public health problem in developed countries.[10][34] M. canetti is rare and seems to be limited to the Horn of Africa, although a few cases have been seen in African emigrants.[35][36] M. microti is also rare and is seen almost only in immunodeficient people, although its prevalence may be significantly underestimated.[37]
Other known pathogenic mycobacteria include M. leprae, M. avium, and M. kansasii. The latter two species are classified as "nontuberculous mycobacteria" (NTM). NTM cause neither TB nor leprosy, but they do cause pulmonary diseases that resemble TB.[38]
Risk factors
A number of factors make people more susceptible to TB infections. The most important risk factor globally is HIV; 13% of all people with TB are infected by the virus.[39] This is a particular problem in sub-Saharan Africa, where rates of HIV are high.[40][41] Of people without HIV who are infected with tuberculosis, about 5–10% develop active disease during their lifetimes;[13] in contrast, 30% of those coinfected with HIV develop the active disease.[13]
Tuberculosis is closely linked to both overcrowding and malnutrition, making it one of the principal diseases of poverty.[11] Those at high risk thus include: people who inject illicit drugs, inhabitants and employees of locales where vulnerable people gather (e.g. prisons and homeless shelters), medically underprivileged and resource-poor communities, high-risk ethnic minorities, children in close contact with high-risk category patients, and health-care providers serving these patients.[42]
Chronic lung disease is another significant risk factor. Silicosis increases the risk about 30-fold.[43] Those who smoke cigarettes have nearly twice the risk of TB compared to nonsmokers.[44]
Other disease states can also increase the risk of developing tuberculosis. These include alcoholism[11] and diabetes mellitus (three-fold increase).[45]
Certain medications, such as corticosteroids and infliximab (an anti-αTNF monoclonal antibody), are becoming increasingly important risk factors, especially in the developed world.[11]
Genetic susceptibility also exists,[46] for which the overall importance remains undefined.[11]
Mechanism

Transmission
When people with active pulmonary TB cough, sneeze, speak, sing, or spit, they expel infectious aerosol droplets 0.5 to 5.0 µm in diameter. A single sneeze can release up to 40,000 droplets.[47] Each one of these droplets may transmit the disease, since the infectious dose of tuberculosis is very small (the inhalation of fewer than 10 bacteria may cause an infection).[48]
People with prolonged, frequent, or close contact with people with TB are at particularly high risk of becoming infected, with an estimated 22% infection rate.[49] A person with active but untreated tuberculosis may infect 10–15 (or more) other people per year.[50] Transmission should occur from only people with active TB – those with latent infection are not thought to be contagious.[10] The probability of transmission from one person to another depends upon several factors, including the number of infectious droplets expelled by the carrier, the effectiveness of ventilation, the duration of exposure, the virulence of the M. tuberculosis strain, the level of immunity in the uninfected person, and others.[51] The cascade of person-to-person spread can be circumvented by segregating those with active ("overt") TB and putting them on anti-TB drug regimens. After about two weeks of effective treatment, subjects with nonresistant active infections generally do not remain contagious to others.[49] If someone does become infected, it typically takes three to four weeks before the newly infected person becomes infectious enough to transmit the disease to others.[52]
Pathogenesis
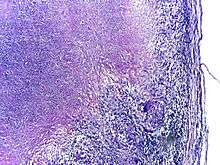
About 90% of those infected with M. tuberculosis have asymptomatic, latent TB infections (sometimes called LTBI),[53] with only a 10% lifetime chance that the latent infection will progress to overt, active tuberculous disease.[54] In those with HIV, the risk of developing active TB increases to nearly 10% a year.[54] If effective treatment is not given, the death rate for active TB cases is up to 66%.[50]
TB infection begins when the mycobacteria reach the pulmonary alveoli, where they invade and replicate within endosomes of alveolar macrophages.[10][55] Macrophages identify the bacterium as foreign and attempt to eliminate it by phagocytosis. During this process, the bacterium is enveloped by the macrophage and stored temporarily in a membrane-bound vesicle called a phagosome. The phagosome then combines with a lysosome to create a phagolysosome. In the phagolysosome, the cell attempts to use reactive oxygen species and acid to kill the bacterium. However, M. tuberculosis has a thick, waxy mycolic acid capsule that protects it from these toxic substances. M. tuberculosis is able to reproduce inside the macrophage and will eventually kill the immune cell.
The primary site of infection in the lungs, known as the "Ghon focus", is generally located in either the upper part of the lower lobe, or the lower part of the upper lobe.[10] Tuberculosis of the lungs may also occur via infection from the blood stream. This is known as a Simon focus and is typically found in the top of the lung.[56] This hematogenous transmission can also spread infection to more distant sites, such as peripheral lymph nodes, the kidneys, the brain, and the bones.[10][57] All parts of the body can be affected by the disease, though for unknown reasons it rarely affects the heart, skeletal muscles, pancreas, or thyroid.[58]
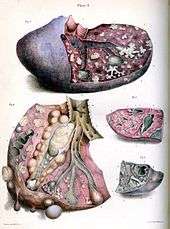
Tuberculosis is classified as one of the granulomatous inflammatory diseases. Macrophages, T lymphocytes, B lymphocytes, and fibroblasts aggregate to form granulomas, with lymphocytes surrounding the infected macrophages. When other macrophages attack the infected macrophage, they fuse together to form a giant multinucleated cell in the alveolar lumen. The granuloma may prevent dissemination of the mycobacteria and provide a local environment for interaction of cells of the immune system.[60] However, more recent evidence suggests that the bacteria use the granulomas to avoid destruction by the host's immune system. Macrophages and dendritic cells in the granulomas are unable to present antigen to lymphocytes; thus the immune response is suppressed.[61] Bacteria inside the granuloma can become dormant, resulting in latent infection. Another feature of the granulomas is the development of abnormal cell death (necrosis) in the center of tubercles. To the naked eye, this has the texture of soft, white cheese and is termed caseous necrosis.[60]
If TB bacteria gain entry to the blood stream from an area of damaged tissue, they can spread throughout the body and set up many foci of infection, all appearing as tiny, white tubercles in the tissues.[62] This severe form of TB disease, most common in young children and those with HIV, is called miliary tuberculosis.[63] People with this disseminated TB have a high fatality rate even with treatment (about 30%).[21][64]
In many people, the infection waxes and wanes. Tissue destruction and necrosis are often balanced by healing and fibrosis.[60] Affected tissue is replaced by scarring and cavities filled with caseous necrotic material. During active disease, some of these cavities are joined to the air passages bronchi and this material can be coughed up. It contains living bacteria, so can spread the infection. Treatment with appropriate antibiotics kills bacteria and allows healing to take place. Upon cure, affected areas are eventually replaced by scar tissue.[60]
Diagnosis

Active tuberculosis
Diagnosing active tuberculosis based only on signs and symptoms is difficult,[65] as is diagnosing the disease in those who are immunosuppressed.[66] A diagnosis of TB should, however, be considered in those with signs of lung disease or constitutional symptoms lasting longer than two weeks.[66] A chest X-ray and multiple sputum cultures for acid-fast bacilli are typically part of the initial evaluation.[66] Interferon-γ release assays and tuberculin skin tests are of little use in the developing world.[67][68] IGRA have similar limitations in those with HIV.[68][69]
A definitive diagnosis of TB is made by identifying M. tuberculosis in a clinical sample (e.g., sputum, pus, or a tissue biopsy). However, the difficult culture process for this slow-growing organism can take two to six weeks for blood or sputum culture.[70] Thus, treatment is often begun before cultures are confirmed.[71]
Nucleic acid amplification tests and adenosine deaminase testing may allow rapid diagnosis of TB.[65] These tests, however, are not routinely recommended, as they rarely alter how a person is treated.[71] Blood tests to detect antibodies are not specific or sensitive, so they are not recommended.[72]
Latent tuberculosis
The Mantoux tuberculin skin test is often used to screen people at high risk for TB.[66] Those who have been previously immunized may have a false-positive test result.[73] The test may be falsely negative in those with sarcoidosis, Hodgkin's lymphoma, malnutrition, and most notably, active tuberculosis.[10] Interferon gamma release assays (IGRAs), on a blood sample, are recommended in those who are positive to the Mantoux test.[71] These are not affected by immunization or most environmental mycobacteria, so they generate fewer false-positive results.[74] However, they are affected by M. szulgai, M. marinum, and M. kansasii.[75] IGRAs may increase sensitivity when used in addition to the skin test, but may be less sensitive than the skin test when used alone.[76]
Prevention
Tuberculosis prevention and control efforts rely primarily on the vaccination of infants and the detection and appropriate treatment of active cases.[11] The World Health Organization has achieved some success with improved treatment regimens, and a small decrease in case numbers.[11] The US Preventive Services Task Force (USPSTF) recommends screening people who are at high risk for latent tuberculosis with either tuberculin skin tests or interferon-gamma release assays.[77]
Vaccines
The only available vaccine as of 2011 is Bacillus Calmette-Guérin (BCG).[78] In children it decreases the risk of getting the infection by 20% and the risk of infection turning into disease by nearly 60%.[79]
It is the most widely used vaccine worldwide, with more than 90% of all children being vaccinated.[11] The immunity it induces decreases after about ten years.[11] As tuberculosis is uncommon in most of Canada, the United Kingdom, and the United States, BCG is administered to only those people at high risk.[80][81][82] Part of the reasoning against the use of the vaccine is that it makes the tuberculin skin test falsely positive, reducing the test's use in screening.[82] A number of new vaccines are currently in development.[11]
Public health
The World Health Organization declared TB a "global health emergency" in 1993,[11] and in 2006, the Stop TB Partnership developed a Global Plan to Stop Tuberculosis that aimed to save 14 million lives between its launch and 2015.[83] A number of targets they set were not achieved by 2015, mostly due to the increase in HIV-associated tuberculosis and the emergence of multiple drug-resistant tuberculosis.[11] A tuberculosis classification system developed by the American Thoracic Society is used primarily in public health programs.[84]
Management
Treatment of TB uses antibiotics to kill the bacteria. Effective TB treatment is difficult, due to the unusual structure and chemical composition of the mycobacterial cell wall, which hinders the entry of drugs and makes many antibiotics ineffective.[85] The two antibiotics most commonly used are isoniazid and rifampicin, and treatments can be prolonged, taking several months.[51] Latent TB treatment usually employs a single antibiotic,[86] while active TB disease is best treated with combinations of several antibiotics to reduce the risk of the bacteria developing antibiotic resistance.[11] People with latent infections are also treated to prevent them from progressing to active TB disease later in life.[86] Directly observed therapy, i.e., having a health care provider watch the person take their medications, is recommended by the WHO in an effort to reduce the number of people not appropriately taking antibiotics.[87] The evidence to support this practice over people simply taking their medications independently is poor.[88] Methods to remind people of the importance of treatment do, however, appear effective.[89]
New onset
The recommended treatment of new-onset pulmonary tuberculosis, as of 2010, is six months of a combination of antibiotics containing rifampicin, isoniazid, pyrazinamide, and ethambutol for the first two months, and only rifampicin and isoniazid for the last four months.[11] Where resistance to isoniazid is high, ethambutol may be added for the last four months as an alternative.[11]
Recurrent disease
If tuberculosis recurs, testing to determine to which antibiotics it is sensitive is important before determining treatment.[11] If multiple drug-resistant TB is detected, treatment with at least four effective antibiotics for 18 to 24 months is recommended.[11]
Medication resistance
Primary resistance occurs when a person becomes infected with a resistant strain of TB. A person with fully susceptible MTB may develop secondary (acquired) resistance during therapy because of inadequate treatment, not taking the prescribed regimen appropriately (lack of compliance), or using low-quality medication.[90] Drug-resistant TB is a serious public health issue in many developing countries, as its treatment is longer and requires more expensive drugs. MDR-TB is defined as resistance to the two most effective first-line TB drugs: rifampicin and isoniazid. Extensively drug-resistant TB is also resistant to three or more of the six classes of second-line drugs.[91] Totally drug-resistant TB is resistant to all currently used drugs.[92] It was first observed in 2003 in Italy,[93] but not widely reported until 2012,[92][94] and has also been found in Iran and India.[95][96] Bedaquiline is tentatively supported for use in multiple drug-resistant TB.[97]
XDR-TB is a term sometimes used to define extensively resistant TB, and constitutes one in ten cases of MDR-TB. Cases of XDR TB have been identified in more than 90% of countries.[95]
Prognosis
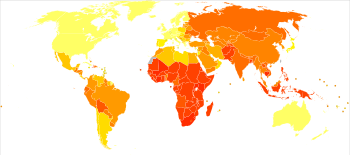
no data
≤10
10–25
25–50
50–75
75–100
100–250
|
250–500
500–750
750–1000
1000–2000
2000–3000
≥ 3000
|
Progression from TB infection to overt TB disease occurs when the bacilli overcome the immune system defenses and begin to multiply. In primary TB disease (some 1–5% of cases), this occurs soon after the initial infection.[10] However, in the majority of cases, a latent infection occurs with no obvious symptoms.[10] These dormant bacilli produce active tuberculosis in 5–10% of these latent cases, often many years after infection.[13]
The risk of reactivation increases with immunosuppression, such as that caused by infection with HIV. In people coinfected with M. tuberculosis and HIV, the risk of reactivation increases to 10% per year.[10] Studies using DNA fingerprinting of M. tuberculosis strains have shown reinfection contributes more substantially to recurrent TB than previously thought,[99] with estimates that it might account for more than 50% of reactivated cases in areas where TB is common.[100] The chance of death from a case of tuberculosis is about 4% as of 2008, down from 8% in 1995.[11]
Epidemiology
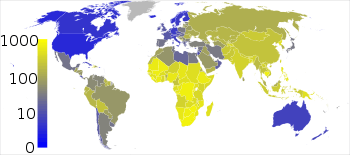
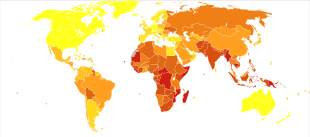
Roughly one-third of the world's population has been infected with M. tuberculosis,[50] with new infections occurring in about 1% of the population each year.[9] However, most infections with M. tuberculosis do not cause TB disease,[102] and 90–95% of infections remain asymptomatic.[53] In 2012, an estimated 8.6 million chronic cases were active.[103] In 2010, 8.8 million new cases of TB were diagnosed, and 1.20–1.45 million deaths occurred, most of these occurring in developing countries.[39][104] Of these 1.45 million deaths, about 0.35 million occur in those also infected with HIV.[105]
Tuberculosis is the second-most common cause of death from infectious disease (after those due to HIV/AIDS).[3] The total number of tuberculosis cases has been decreasing since 2005, while new cases have decreased since 2002.[39] China has achieved particularly dramatic progress, with about an 80% reduction in its TB mortality rate between 1990 and 2010.[105] The number of new cases has declined by 17% between 2004–2014.[95] Tuberculosis is more common in developing countries; about 80% of the population in many Asian and African countries test positive in tuberculin tests, while only 5–10% of the US population test positive.[10] Hopes of totally controlling the disease have been dramatically dampened because of a number of factors, including the difficulty of developing an effective vaccine, the expensive and time-consuming diagnostic process, the necessity of many months of treatment, the increase in HIV-associated tuberculosis, and the emergence of drug-resistant cases in the 1980s.[11]
In 2007, the country with the highest estimated incidence rate of TB was Swaziland, with 1,200 cases per 100,000 people. India had the largest total incidence, with an estimated 2.0 million new cases.[106] In developed countries, tuberculosis is less common and is found mainly in urban areas. Rates per 100,000 people in different areas of the world were: globally 178, Africa 332, the Americas 36, Eastern Mediterranean 173, Europe 63, Southeast Asia 278, and Western Pacific 139 in 2010.[105] In Canada and Australia, tuberculosis is many times more common among the aboriginal peoples, especially in remote areas.[107][108] In the United States Native Americans have a fivefold greater mortality from TB,[109] and racial and ethnic minorities accounted for 84% of all reported TB cases.[110]
The rates of TB varies with age. In Africa, it primarily affects adolescents and young adults.[111] However, in countries where incidence rates have declined dramatically (such as the United States), TB is mainly a disease of older people and the immunocompromised (risk factors are listed above).[10][112] Worldwide, 22 "high-burden" states or countries together experience 80% of cases as well as 83% of deaths.[95]
History

Tuberculosis has been present in humans since antiquity.[11] The earliest unambiguous detection of M. tuberculosis involves evidence of the disease in the remains of bison in Wyoming dated to around 17,000 years ago.[113] However, whether tuberculosis originated in bovines, then was transferred to humans, or whether it diverged from a common ancestor, is currently unclear.[114] A comparison of the genes of M. tuberculosis complex (MTBC) in humans to MTBC in animals suggests humans did not acquire MTBC from animals during animal domestication, as was previously believed. Both strains of the tuberculosis bacteria share a common ancestor, which could have infected humans even before the Neolithic Revolution.[115]
Skeletal remains show prehistoric humans (4000 BC) had TB, and researchers have found tubercular decay in the spines of Egyptian mummies dating from 3000–2400 BC.[116] Genetic studies suggest TB was present in the Americas from about 100 AD.[117]
Before the Industrial Revolution, folklore often associated tuberculosis with vampires. When one member of a family died from it, the other infected members would lose their health slowly. People believed this was caused by the original person with TB draining the life from the other family members.[118]
Although the pulmonary form associated with tubercles was established as a pathology by Dr Richard Morton in 1689,[119][120] due to the variety of its symptoms, TB was not identified as a single disease until the 1820s. It was not named "tuberculosis" until 1839, by J. L. Schönlein.[121]
During 1838–1845, Dr. John Croghan, the owner of Mammoth Cave, brought a number of people with tuberculosis into the cave in the hope of curing the disease with the constant temperature and purity of the cave air; they died within a year.[122] Hermann Brehmer opened the first TB sanatorium in 1859 in Görbersdorf (now Sokołowsko), Silesia.[123]

The bacillus causing tuberculosis, M. tuberculosis, was identified and described on 24 March 1882 by Robert Koch. He received the Nobel Prize in physiology or medicine in 1905 for this discovery.[124] Koch did not believe the bovine (cattle) and human tuberculosis diseases were similar, which delayed the recognition of infected milk as a source of infection. Later, the risk of transmission from this source was dramatically reduced by the invention of the pasteurization process. Koch announced a glycerine extract of the tubercle bacilli as a "remedy" for tuberculosis in 1890, calling it "tuberculin". While it was not effective, it was later successfully adapted as a screening test for the presence of pre-symptomatic tuberculosis.[125] The World Tuberculosis Day was established on 24 March for this reason.
Albert Calmette and Camille Guérin achieved the first genuine success in immunization against tuberculosis in 1906, using attenuated bovine-strain tuberculosis. It was called bacille Calmette–Guérin (BCG). The BCG vaccine was first used on humans in 1921 in France,[126] but received widespread acceptance in the US, Great Britain, and Germany only after World War II.[127]
Tuberculosis caused the most widespread public concern in the 19th and early 20th centuries as an endemic disease of the urban poor. In 1815, one in four deaths in England was due to "consumption". By 1918, one in six deaths in France was still caused by TB. After TB was determined to be contagious, in the 1880s, it was put on a notifiable disease list in Britain; campaigns were started to stop people from spitting in public places, and the infected poor were "encouraged" to enter sanatoria that resembled prisons (the sanatoria for the middle and upper classes offered excellent care and constant medical attention).[123] Whatever the (purported) benefits of the "fresh air" and labor in the sanatoria, even under the best conditions, 50% of those who entered died within five years (circa 1916).[123]
In Europe, rates of tuberculosis began to rise in the early 1600s to a peak level in the 1800s, when it caused nearly 25% of all deaths.[128] By the 1950s, mortality had decreased nearly 90%.[129] Improvements in public health began significantly reducing rates of tuberculosis even before the arrival of streptomycin and other antibiotics, although the disease remained a significant threat to public health such that when the Medical Research Council was formed in Britain in 1913, its initial focus was tuberculosis research.[130]
In 1946, the development of the antibiotic streptomycin made effective treatment and cure of TB a reality. Prior to the introduction of this drug, the only treatment (except sanatoria) was surgical intervention, including the "pneumothorax technique", which involved collapsing an infected lung to "rest" it and allow tuberculous lesions to heal.[131]
Because of the emergence of MDR-TB, surgery has been re-introduced as an option within the generally accepted standard of care in treating TB infections. Current surgical interventions involve removal of pathological chest cavities ("bullae") in the lungs to reduce the number of bacteria and to increase the exposure of the remaining bacteria to drugs in the bloodstream, thereby simultaneously reducing the total bacterial load and increasing the effectiveness of systemic antibiotic therapy.[132]
Hopes of completely eliminating TB (cf. smallpox) from the population were dashed after the rise of drug-resistant strains in the 1980s. The subsequent resurgence of tuberculosis resulted in the declaration of a global health emergency by the World Health Organization in 1993.[133]
Society and culture
Names
Phthisis (Φθισις) is a Greek word for consumption, an old term for pulmonary tuberculosis;[2] around 460 BC, Hippocrates described phthisis as a disease of dry seasons.[134] The abbreviation "TB" is short for tubercle bacillus.
"Consumption" was the most common nineteenth century English word for the disease. The Latin root "con" meaning "completely" is linked to "sumere" meaning "to take up from under."[135] In The Life and Death of Mr. Badman by John Bunyan, the author calls consumption "the captain of all these men of death."[136]
Public health efforts
The World Health Organization, Bill and Melinda Gates Foundation, and US government are subsidizing a fast-acting diagnostic tuberculosis test for use in low- and middle-income countries.[137][138][139] In addition to being fast-acting, the test can determine if there is resistance to the antibiotic rifampicin which may indicate multi-drug resistant tuberculosis and is accurate in those who are also infected with HIV.[137][140] Many resource-poor places as of 2011 have access to only sputum microscopy.[141]
India had the highest total number of TB cases worldwide in 2010, in part due to poor disease management within the private and public health care sector.[142] Programs such as the Revised National Tuberculosis Control Program are working to reduce TB levels amongst people receiving public health care.[143][144]
A 2014 the EIU-healthcare report that the need to address apathy and urging for increased funding. The report cites among others Lucica Ditui "[TB] is like an orphan. It has been neglected even in countries with a high burden and often forgotten by donors and those investing in health interventions."[95]
Slow progress has led to frustration, expressed by the executive director of the Global Fund to Fight AIDS, Tuberculosis and Malaria – Mark Dybul: "we have the tools to end TB as a pandemic and public health threat on the planet, but we are not doing it."[95] Several international organizations are pushing for more transparency in treatment, and more countries are implementing mandatory reporting of cases to the government, although adherence is often sketchy. Commercial treatment providers may at times overprescribe second-line drugs as well as supplementary treatment, promoting demands for further regulations.[95] The government of Brazil provides universal TB-care, which reduces this problem.[95] Conversely, falling rates of TB-infection may not relate to the number of programs directed at reducing infection rates, but may be tied to increased level of education, income, and health of the population.[95] Costs of the disease, as calculated by the World Bank in 2009 may exceed 150 billion USD per year in "high burden" countries.[95] Lack of progress eradicating the disease may also be due to lack of patient follow-up – as among the 250M rural migrants in China.[95]
Stigma
Slow progress in preventing the disease may in part be due to stigma associated with TB.[95] Stigma may be due to the fear of transmission from affected individuals. This stigma may additionally arise due to links between TB and poverty, and in Africa, AIDS.[95] Such stigmatization may be both real and perceived, for example; in Ghana individuals with TB are banned from attending public gatherings.[145]
Stigma towards TB may result in delays in seeking treatment,[95] lower treatment compliance, and family members keeping cause of death secret[145] – allowing the disease to spread further.[95] At odds is Russia, where stigma was associated with increased treatment compliance.[145] TB stigma also affects socially marginalized individuals to a greater degree and varies between regions.[145]
One way to decrease stigma may be through the promotion of "TB clubs", where those infected may share experiences and offer support, or through counseling.[145] Some studies have shown TB education programs to be effective in decreasing stigma, and may thus be effective in increasing treatment adherence.[145] Despite this, studies on the relationship between reduced stigma and mortality are lacking as of 2010, and similar efforts to decrease stigma surrounding AIDS have been minimally effective.[145] Some have claimed the stigma to be worse than the disease, and healthcare providers may unintentionally reinforce stigma, as those with TB are often perceived as difficult or otherwise undesirable.[95] A greater understanding of the social and cultural dimensions of tuberculosis may also help with stigma reduction.[146]
Research
The BCG vaccine has limitations, and research to develop new TB vaccines is ongoing.[147] A number of potential candidates are currently in phase I and II clinical trials.[147] Two main approaches are being used to attempt to improve the efficacy of available vaccines. One approach involves adding a subunit vaccine to BCG, while the other strategy is attempting to create new and better live vaccines.[147] MVA85A, an example of a subunit vaccine, currently in trials in South Africa, is based on a genetically modified vaccinia virus.[148] Vaccines are hoped to play a significant role in treatment of both latent and active disease.[149]
To encourage further discovery, researchers and policymakers are promoting new economic models of vaccine development, including prizes, tax incentives, and advance market commitments.[150][151] A number of groups, including the Stop TB Partnership,[152] the South African Tuberculosis Vaccine Initiative, and the Aeras Global TB Vaccine Foundation, are involved with research.[153] Among these, the Aeras Global TB Vaccine Foundation received a gift of more than $280 million (US) from the Bill and Melinda Gates Foundation to develop and license an improved vaccine against tuberculosis for use in high burden countries.[154][155]
A number of medications are being studied for multi drug resistant tuberculosis including: bedaquiline and delamanid.[156] Bedaquiline received U.S. Food and Drug Administration (FDA) approval in late 2012.[157] The safety and effectiveness of these new agents are still uncertain, because they are based on the results of a relatively small studies.[156][158] However, existing data suggest that patients taking bedaquiline in addition to standard TB therapy are five times more likely to die than those without the new drug,[159] which has resulted in medical journal articles raising health policy questions about why the FDA approved the drug and whether financial ties to the company making bedaquiline influenced physicians' support for its use [158][160]
Other animals
Mycobacteria infect many different animals, including birds,[161] rodents,[162] and reptiles.[163] The subspecies Mycobacterium tuberculosis, though, is rarely present in wild animals.[164] An effort to eradicate bovine tuberculosis caused by Mycobacterium bovis from the cattle and deer herds of New Zealand has been relatively successful.[165] Efforts in Great Britain have been less successful.[166][167]
As of 2015, tuberculosis appears to be widespread among captive elephants in the US. It is believed that the animals originally acquired the disease from humans, a process called reverse zoonosis. Because the disease can spread through the air to infect both humans and other animals, it is a public health concern affecting circuses and zoos.[168][169]
References
- 1 2 3 4 5 6 7 8 "Tuberculosis Fact sheet N°104". WHO. October 2015. Retrieved 11 February 2016.
- 1 2 The Chambers Dictionary. New Delhi: Allied Chambers India Ltd. 1998. p. 352. ISBN 978-81-86062-25-8.
- 1 2 3 4 5 6 7 8 9 10 Dolin, [edited by] Gerald L. Mandell, John E. Bennett, Raphael (2010). Mandell, Douglas, and Bennett's principles and practice of infectious diseases (7th ed.). Philadelphia, PA: Churchill Livingstone/Elsevier. pp. Chapter 250. ISBN 978-0-443-06839-3.
- ↑ "Basic TB Facts". CDC. March 13, 2012. Retrieved 11 February 2016.
- ↑ Konstantinos A (2010). "Testing for tuberculosis". Australian Prescriber. 33 (1): 12–18.
- ↑ Hawn, TR; Day, TA; Scriba, TJ; Hatherill, M; Hanekom, WA; Evans, TG; Churchyard, GJ; Kublin, JG; Bekker, LG; Self, SG (December 2014). "Tuberculosis vaccines and prevention of infection.". Microbiology and molecular biology reviews: MMBR. 78 (4): 650–71. doi:10.1128/MMBR.00021-14. PMC 4248657
 . PMID 25428938.
. PMID 25428938. - ↑ Harris, Randall E. (2013). Epidemiology of chronic disease: global perspectives. Burlington, MA: Jones & Bartlett Learning. p. 682. ISBN 9780763780470.
- 1 2 Organization, World Health (2008). Implementing the WHO Stop TB Strategy: a handbook for national TB control programmes. Geneva: World Health Organization. p. 179. ISBN 9789241546676.
- 1 2 "Tuberculosis". World Health Organization. 2002.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 Kumar V, Abbas AK, Fausto N, Mitchell RN (2007). Robbins Basic Pathology (8th ed.). Saunders Elsevier. pp. 516–522. ISBN 978-1-4160-2973-1.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 Lawn, SD; Zumla, AI (2 July 2011). "Tuberculosis". Lancet. 378 (9785): 57–72. doi:10.1016/S0140-6736(10)62173-3. PMID 21420161.
- ↑ Schiffman G (15 January 2009). "Tuberculosis Symptoms". eMedicineHealth.
- 1 2 3 4 Gibson, Peter G. (ed.); Abramson, Michael (ed.); Wood-Baker, Richard (ed.); Volmink, Jimmy (ed.); Hensley, Michael (ed.); Costabel, Ulrich (ed.) (2005). Evidence-Based Respiratory Medicine (1st ed.). BMJ Books. p. 321. ISBN 978-0-7279-1605-1.
- ↑ Behera, D. (2010). Textbook of Pulmonary Medicine (2nd ed.). New Delhi: Jaypee Brothers Medical Publishers. p. 457. ISBN 978-81-8448-749-7.
- ↑ Halezeroğlu, S; Okur, E (March 2014). "Thoracic surgery for haemoptysis in the context of tuberculosis: what is the best management approach?". Journal of Thoracic Disease. 6 (3): 182–5. doi:10.3978/j.issn.2072-1439.2013.12.25. PMID 24624281.
- ↑ Jindal, editor-in-chief SK (2011). Textbook of Pulmonary and Critical Care Medicine. New Delhi: Jaypee Brothers Medical Publishers. p. 549. ISBN 978-93-5025-073-0.
- 1 2 Golden MP, Vikram HR (2005). "Extrapulmonary tuberculosis: an overview". American Family Physician. 72 (9): 1761–8. PMID 16300038.
- ↑ Kabra, [edited by] Vimlesh Seth, S.K. (2006). Essentials of tuberculosis in children (3rd ed.). New Delhi: Jaypee Bros. Medical Publishers. p. 249. ISBN 978-81-8061-709-6.
- ↑ Manual of Surgery. Kaplan Publishing. 2008. p. 75. ISBN 9781427797995.
- ↑ Burkitt, H. George (2007). Essential Surgery: Problems, Diagnosis & Management 4th ed. p. 34. ISBN 9780443103452.
- 1 2 Ghosh, editors-in-chief, Thomas M. Habermann, Amit K. (2008). Mayo Clinic internal medicine: concise textbook. Rochester, MN: Mayo Clinic Scientific Press. p. 789. ISBN 978-1-4200-6749-1.
- ↑ Southwick F (10 December 2007). "Chapter 4: Pulmonary Infections". Infectious Diseases: A Clinical Short Course, 2nd ed. McGraw-Hill Medical Publishing Division. pp. 104, 313–4. ISBN 0-07-147722-5.
- ↑ Jindal, editor-in-chief SK (2011). Textbook of Pulmonary and Critical Care Medicine. New Delhi: Jaypee Brothers Medical Publishers. p. 525. ISBN 978-93-5025-073-0.
- ↑ Niederweis M, Danilchanka O, Huff J, Hoffmann C, Engelhardt H (March 2010). "Mycobacterial outer membranes: in search of proteins". Trends in Microbiology. 18 (3): 109–16. doi:10.1016/j.tim.2009.12.005. PMC 2931330
 . PMID 20060722.
. PMID 20060722. - 1 2 Madison B (2001). "Application of stains in clinical microbiology". Biotechnic & Histochemistry. 76 (3): 119–25. doi:10.1080/714028138. PMID 11475314.
- ↑ Parish T.; Stoker N. (1999). "Mycobacteria: bugs and bugbears (two steps forward and one step back)". Molecular Biotechnology. 13 (3): 191–200. doi:10.1385/MB:13:3:191. PMID 10934532.
- ↑ Medical Laboratory Science: Theory and Practice. New Delhi: Tata McGraw-Hill. 2000. p. 473. ISBN 0-07-463223-X.
- ↑ "Acid-Fast Stain Protocols". 21 August 2013. Retrieved 26 March 2016.
- ↑ Kommareddi S.; Abramowsky C.; Swinehart G.; Hrabak L. (1984). "Nontuberculous mycobacterial infections: comparison of the fluorescent auramine-O and Ziehl-Neelsen techniques in tissue diagnosis". Human Pathology. 15 (11): 1085–1089. doi:10.1016/S0046-8177(84)80253-1. PMID 6208117.
- ↑ van Lettow, Monique; Whalen, Christopher (2008). Nutrition and health in developing countries (2nd ed.). Totowa, N.J. (Richard D. Semba and Martin W. Bloem, eds.): Humana Press. p. 291. ISBN 978-1-934115-24-4.
- ↑ van Soolingen D., et al. (1997). "A novel pathogenic taxon of the Mycobacterium tuberculosis complex, Canetti: characterization of an exceptional isolate from Africa". International Journal of Systematic Bacteriology. 47 (4): 1236–45. doi:10.1099/00207713-47-4-1236. PMID 9336935.
- ↑ Niemann S., et al. (2002). "Mycobacterium africanum Subtype II Is Associated with Two Distinct Genotypes and Is a Major Cause of Human Tuberculosis in Kampala, Uganda". Journal of Clinical Microbiology. 40 (9): 3398–405. doi:10.1128/JCM.40.9.3398-3405.2002. PMC 130701
 . PMID 12202584.
. PMID 12202584. - ↑ Niobe-Eyangoh S.N., et al. (2003). "Genetic Biodiversity of Mycobacterium tuberculosis Complex Strains from Patients with Pulmonary Tuberculosis in Cameroon". Journal of Clinical Microbiology. 41 (6): 2547–53. doi:10.1128/JCM.41.6.2547-2553.2003. PMC 156567
 . PMID 12791879.
. PMID 12791879. - ↑ Thoen C, Lobue P, de Kantor I (2006). "The importance of Mycobacterium bovis as a zoonosis". Veterinary Microbiology. 112 (2–4): 339–45. doi:10.1016/j.vetmic.2005.11.047. PMID 16387455.
- ↑ Acton, Q. Ashton (2011). Mycobacterium Infections: New Insights for the Healthcare Professional. ScholarlyEditions. p. 1968. ISBN 978-1-4649-0122-5.
- ↑ Pfyffer, GE; Auckenthaler, R; van Embden, JD; van Soolingen, D (Oct–Dec 1998). "Mycobacterium canettii, the smooth variant of M. tuberculosis, isolated from a Swiss patient exposed in Africa". Emerging Infectious Diseases. 4 (4): 631–4. doi:10.3201/eid0404.980414. PMC 2640258
 . PMID 9866740.
. PMID 9866740. - ↑ Panteix, G; Gutierrez, MC; Boschiroli, ML; Rouviere, M; Plaidy, A; Pressac, D; Porcheret, H; Chyderiotis, G; Ponsada, M; Van Oortegem, K; Salloum, S; Cabuzel, S; Bañuls, AL; Van de Perre, P; Godreuil, S (August 2010). "Pulmonary tuberculosis due to Mycobacterium microti: a study of six recent cases in France". Journal of Medical Microbiology. 59 (Pt 8): 984–9. doi:10.1099/jmm.0.019372-0. PMID 20488936.
- ↑ American Thoracic Society (1997). "Diagnosis and treatment of disease caused by nontuberculous mycobacteria. This official statement of the American Thoracic Society was approved by the Board of Directors, March 1997. Medical Section of the American Lung Association". American Journal of Respiratory and Critical Care Medicine. 156 (2 Pt 2): S1–25. doi:10.1164/ajrccm.156.2.atsstatement. PMID 9279284.
- 1 2 3 World Health Organization (2011). "The sixteenth global report on tuberculosis" (PDF).
- ↑ World Health Organization. "Global tuberculosis control–surveillance, planning, financing WHO Report 2006". Retrieved 13 October 2006.
- ↑ Chaisson, RE; Martinson, NA (13 March 2008). "Tuberculosis in Africa—combating an HIV-driven crisis". The New England Journal of Medicine. 358 (11): 1089–92. doi:10.1056/NEJMp0800809. PMID 18337598.
- ↑ Griffith D, Kerr C (1996). "Tuberculosis: disease of the past, disease of the present". Journal of Perianesthesia Nursing. 11 (4): 240–5. doi:10.1016/S1089-9472(96)80023-2. PMID 8964016.
- ↑ ATS/CDC Statement Committee on Latent Tuberculosis Infection (June 2000). "Targeted tuberculin testing and treatment of latent tuberculosis infection. American Thoracic Society". MMWR. Recommendations and Reports. 49 (RR–6): 1–51. PMID 10881762.
- ↑ van Zyl Smit, RN; Pai, M; Yew, WW; Leung, CC; Zumla, A; Bateman, ED; Dheda, K (January 2010). "Global lung health: the colliding epidemics of tuberculosis, tobacco smoking, HIV and COPD". European Respiratory Journal. 35 (1): 27–33. doi:10.1183/09031936.00072909. PMID 20044459.
These analyses indicate that smokers are almost twice as likely to be infected with TB and to progress to active disease (RR of about 1.5 for latent TB infection (LTBI) and RR of ∼2.0 for TB disease). Smokers are also twice as likely to die from TB (RR of about 2.0 for TB mortality), but data are difficult to interpret because of heterogeneity in the results across studies.
- ↑ Restrepo, BI (15 August 2007). "Convergence of the tuberculosis and diabetes epidemics: renewal of old acquaintances". Clinical Infectious Diseases. 45 (4): 436–8. doi:10.1086/519939. PMC 2900315
 . PMID 17638190.
. PMID 17638190. - ↑ Möller, M; Hoal, EG (March 2010). "Current findings, challenges and novel approaches in human genetic susceptibility to tuberculosis". Tuberculosis. 90 (2): 71–83. doi:10.1016/j.tube.2010.02.002. PMID 20206579.
- ↑ Cole E, Cook C (1998). "Characterization of infectious aerosols in health care facilities: an aid to effective engineering controls and preventive strategies". Am J Infect Control. 26 (4): 453–64. doi:10.1016/S0196-6553(98)70046-X. PMID 9721404.
- ↑ Nicas M, Nazaroff WW, Hubbard A (2005). "Toward understanding the risk of secondary airborne infection: emission of respirable pathogens". J Occup Environ Hyg. 2 (3): 143–54. doi:10.1080/15459620590918466. PMID 15764538.
- 1 2 Ahmed N, Hasnain S (2011). "Molecular epidemiology of tuberculosis in India: Moving forward with a systems biology approach". Tuberculosis. 91 (5): 407–3. doi:10.1016/j.tube.2011.03.006. PMID 21514230.
- 1 2 3 "Tuberculosis Fact sheet N°104". World Health Organization. November 2010. Retrieved 26 July 2011.
- 1 2 "Core Curriculum on Tuberculosis: What the Clinician Should Know" (PDF) (5th ed.). Centers for Disease Control and Prevention (CDC), Division of Tuberculosis Elimination. 2011. p. 24.
- ↑ "Causes of Tuberculosis". Mayo Clinic. 21 December 2006. Retrieved 19 October 2007.
- 1 2 Skolnik, Richard (2011). Global health 101 (2nd ed.). Burlington, MA: Jones & Bartlett Learning. p. 253. ISBN 978-0-7637-9751-5.
- 1 2 editors, Arch G. Mainous III, Claire Pomeroy, (2009). Management of antimicrobials in infectious diseases: impact of antibiotic resistance. (2nd rev. ed.). Totowa, N.J.: Humana Press. p. 74. ISBN 978-1-60327-238-4.
- ↑ Houben E, Nguyen L, Pieters J (2006). "Interaction of pathogenic mycobacteria with the host immune system". Curr Opin Microbiol. 9 (1): 76–85. doi:10.1016/j.mib.2005.12.014. PMID 16406837.
- ↑ Khan (2011). Essence Of Paediatrics. Elsevier India. p. 401. ISBN 978-81-312-2804-3.
- ↑ Herrmann J, Lagrange P (2005). "Dendritic cells and Mycobacterium tuberculosis: which is the Trojan horse?". Pathol Biol (Paris). 53 (1): 35–40. doi:10.1016/j.patbio.2004.01.004. PMID 15620608.
- ↑ Agarwal R, Malhotra P, Awasthi A, Kakkar N, Gupta D (2005). "Tuberculous dilated cardiomyopathy: an under-recognized entity?". BMC Infect Dis. 5 (1): 29. doi:10.1186/1471-2334-5-29. PMC 1090580
 . PMID 15857515.
. PMID 15857515. - ↑ John Mason Good; Samuel Cooper; Augustus Sidney Doane (1835). The Study of Medicine. Harper. p. 32.
- 1 2 3 4 Grosset J (2003). "Mycobacterium tuberculosis in the Extracellular Compartment: an Underestimated Adversary". Antimicrob Agents Chemother. 47 (3): 833–6. doi:10.1128/AAC.47.3.833-836.2003. PMC 149338
 . PMID 12604509.
. PMID 12604509. - ↑ Bozzano F (2014). "Immunology of tuberculosis". Mediterr J Hematol Infect Dis. 6 (1): e2014027. doi:10.4084/MJHID.2014.027. PMC 4010607
 . PMID 24804000.
. PMID 24804000. - ↑ Crowley, Leonard V. (2010). An introduction to human disease: pathology and pathophysiology correlations (8th ed.). Sudbury, Mass.: Jones and Bartlett. p. 374. ISBN 978-0-7637-6591-0.
- ↑ Anthony, Harries (2005). TB/HIV a Clinical Manual. (2nd ed.). Geneva: World Health Organization. p. 75. ISBN 978-92-4-154634-8.
- ↑ Jacob, JT; Mehta, AK; Leonard, MK (January 2009). "Acute forms of tuberculosis in adults". The American Journal of Medicine. 122 (1): 12–7. doi:10.1016/j.amjmed.2008.09.018. PMID 19114163.
- 1 2 Bento, J; Silva, AS; Rodrigues, F; Duarte, R (Jan–Feb 2011). "[Diagnostic tools in tuberculosis]". Acta Médica Portuguesa. 24 (1): 145–54. PMID 21672452.
- 1 2 3 4 Escalante, P (2 June 2009). "In the clinic. Tuberculosis". Annals of Internal Medicine. 150 (11): ITC61–614; quiz ITV616. doi:10.7326/0003-4819-150-11-200906020-01006. PMID 19487708.
- ↑ Metcalfe, JZ; Everett, CK; Steingart, KR; Cattamanchi, A; Huang, L; Hopewell, PC; Pai, M (15 November 2011). "Interferon-γ release assays for active pulmonary tuberculosis diagnosis in adults in low- and middle-income countries: systematic review and meta-analysis". The Journal of Infectious Diseases. 204 Suppl 4 (suppl_4): S1120–9. doi:10.1093/infdis/jir410. PMC 3192542
 . PMID 21996694.
. PMID 21996694. - 1 2 Sester, M; Sotgiu, G; Lange, C; Giehl, C; Girardi, E; Migliori, GB; Bossink, A; Dheda, K; Diel, R; Dominguez, J; Lipman, M; Nemeth, J; Ravn, P; Winkler, S; Huitric, E; Sandgren, A; Manissero, D (January 2011). "Interferon-γ release assays for the diagnosis of active tuberculosis: a systematic review and meta-analysis". The European Respiratory Journal. 37 (1): 100–11. doi:10.1183/09031936.00114810. PMID 20847080.
- ↑ Chen, J; Zhang, R; Wang, J; Liu, L; Zheng, Y; Shen, Y; Qi, T; Lu, H (2011). Vermund, Sten H, ed. "Interferon-gamma release assays for the diagnosis of active tuberculosis in HIV-infected patients: a systematic review and meta-analysis". PLoS ONE. 6 (11): e26827. doi:10.1371/journal.pone.0026827. PMC 3206065
 . PMID 22069472.
. PMID 22069472. 
- ↑ Diseases, Special Programme for Research & Training in Tropical (2006). Diagnostics for tuberculosis: global demand and market potential. Geneva: World Health Organization on behalf of the Special Programme for Research and Training in Tropical Diseases. p. 36. ISBN 978-92-4-156330-7.
- 1 2 3 National Institute for Health and Clinical Excellence. Clinical guideline 117: Tuberculosis. London, 2011.
- ↑ Steingart, KR; Flores, LL; Dendukuri, N; Schiller, I; Laal, S; Ramsay, A; Hopewell, PC; Pai, M (August 2011). Evans, Carlton, ed. "Commercial serological tests for the diagnosis of active pulmonary and extrapulmonary tuberculosis: an updated systematic review and meta-analysis". PLOS Medicine. 8 (8): e1001062. doi:10.1371/journal.pmed.1001062. PMC 3153457
 . PMID 21857806.
. PMID 21857806. 
- ↑ Rothel J, Andersen P (2005). "Diagnosis of latent Mycobacterium tuberculosis infection: is the demise of the Mantoux test imminent?". Expert Rev Anti Infect Ther. 3 (6): 981–93. doi:10.1586/14787210.3.6.981. PMID 16307510.
- ↑ Pai M, Zwerling A, Menzies D (2008). "Systematic Review: T-Cell–based Assays for the Diagnosis of Latent Tuberculosis Infection: An Update". Ann. Intern. Med. 149 (3): 1–9. doi:10.7326/0003-4819-149-3-200808050-00241. PMC 2951987
 . PMID 18593687.
. PMID 18593687. - ↑ Jindal, editor-in-chief SK (2011). Textbook of Pulmonary and Critical Care Medicine. New Delhi: Jaypee Brothers Medical Publishers. p. 544. ISBN 978-93-5025-073-0.
- ↑ Amicosante, M; Ciccozzi, M; Markova, R (April 2010). "Rational use of immunodiagnostic tools for tuberculosis infection: guidelines and cost effectiveness studies". The new microbiologica. 33 (2): 93–107. PMID 20518271.
- ↑ Bibbins-Domingo, Kirsten; Grossman, David C.; Curry, Susan J.; Bauman, Linda; Davidson, Karina W.; Epling, John W.; García, Francisco A.R.; Herzstein, Jessica; Kemper, Alex R.; Krist, Alex H.; Kurth, Ann E.; Landefeld, C. Seth; Mangione, Carol M.; Phillips, William R.; Phipps, Maureen G.; Pignone, Michael P. (6 September 2016). "Screening for Latent Tuberculosis Infection in Adults". JAMA. 316 (9): 962. doi:10.1001/jama.2016.11046.
- ↑ McShane, H (12 October 2011). "Tuberculosis vaccines: beyond bacille Calmette–Guérin". Philosophical transactions of the Royal Society of London. Series B, Biological sciences. 366 (1579): 2782–9. doi:10.1098/rstb.2011.0097. PMC 3146779
 . PMID 21893541.
. PMID 21893541. - ↑ Roy, A; Eisenhut, M; Harris, RJ; Rodrigues, LC; Sridhar, S; Habermann, S; Snell, L; Mangtani, P; Adetifa, I; Lalvani, A; Abubakar, I (Aug 5, 2014). "Effect of BCG vaccination against Mycobacterium tuberculosis infection in children: systematic review and meta-analysis.". BMJ (Clinical research ed.). 349: g4643. doi:10.1136/bmj.g4643. PMC 4122754
 . PMID 25097193.
. PMID 25097193. - ↑ "Vaccine and Immunizations: TB Vaccine (BCG)". Centers for Disease Control and Prevention. 2011. Retrieved 26 July 2011.
- ↑ "BCG Vaccine Usage in Canada – Current and Historical". Public Health Agency of Canada. September 2010. Retrieved 30 December 2011.
- 1 2 Teo, SS; Shingadia, DV (June 2006). "Does BCG have a role in tuberculosis control and prevention in the United Kingdom?". Archives of Disease in Childhood. 91 (6): 529–31. doi:10.1136/adc.2005.085043. PMC 2082765
 . PMID 16714729.
. PMID 16714729. - ↑ "The Global Plan to Stop TB". World Health Organization. 2011. Retrieved 13 June 2011.
- ↑ Warrell, ed. by D. J. Weatherall ... [4. + 5. ed.] ed. by David A. (2005). Sections 1 – 10. (4. ed., paperback. ed.). Oxford [u.a.]: Oxford Univ. Press. p. 560. ISBN 978-0-19-857014-1.
- ↑ Brennan PJ, Nikaido H (1995). "The envelope of mycobacteria". Annu. Rev. Biochem. 64: 29–63. doi:10.1146/annurev.bi.64.070195.000333. PMID 7574484.
- 1 2 Menzies, D; Al Jahdali, H; Al Otaibi, B (March 2011). "Recent developments in treatment of latent tuberculosis infection". The Indian journal of medical research. 133 (3): 257–66. PMC 3103149
 . PMID 21441678.
. PMID 21441678. - ↑ Arch G.; III Mainous (2010). Management of Antimicrobials in Infectious Diseases: Impact of Antibiotic Resistance. Totowa, N.J.: Humana Press. p. 69. ISBN 1-60327-238-0.
- ↑ Volmink J, Garner P (2007). Volmink, Jimmy, ed. "Directly observed therapy for treating tuberculosis". Cochrane Database Syst Rev (4): CD003343. doi:10.1002/14651858.CD003343.pub3. PMID 17943789.
- ↑ Liu, Q; Abba, K; Alejandria, MM; Balanag, VM; Berba, RP; Lansang, MA (8 October 2008). Liu, Qin, ed. "Reminder systems and late patient tracers in the diagnosis and management of tuberculosis". Cochrane database of systematic reviews (Online) (4): CD006594. doi:10.1002/14651858.CD006594.pub2. PMID 18843723.
- ↑ O'Brien R (1994). "Drug-resistant tuberculosis: etiology, management and prevention". Semin Respir Infect. 9 (2): 104–12. PMID 7973169.
- ↑ Centers for Disease Control and Prevention (CDC) (2006). "Emergence of Mycobacterium tuberculosis with extensive resistance to second-line drugs—worldwide, 2000–2004". MMWR Morb Mortal Wkly Rep. 55 (11): 301–5. PMID 16557213.
- 1 2 Maryn McKenna (12 January 2012). "Totally Resistant TB: Earliest Cases in Italy". Wired. Retrieved 12 January 2012.
- ↑ Migliori, G.B.; De Iaco, G.; Besozzi, G.; Centis, R.; Cirillo, D.M. (May 17, 2007). "First tuberculosis cases in Italy resistant to all tested drugs". Euro surveillance: bulletin Européen sur les maladies transmissibles (European communicable disease bulletin). 12 (5): E070517.1. PMID 17868596.
- ↑ "Totally Drug-Resistant TB: a WHO consultation on the diagnostic definition and treatment options" (PDF). who.int. World Health Organization. Retrieved 25 March 2016.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 Paul Kielstra (30 June 2014). Zoe Tabary, ed. "Ancient enemy, modern imperative – A time for greater action against tuberculosis". Economist Insights. The Economist Group. Retrieved 1 August 2014.
- ↑ Velayati, A.A.; Masjedi, M.R.; Farnia, P.; Tabarsi, P.; Ghanavi, J.; Ziazarifi, A.H.; Hoffner, S.E. (August 2009). "Emergence of new forms of totally drug-resistant tuberculosis bacilli: super extensively drug-resistant tuberculosis or totally drug-resistant strains in Iran". Chest. 136 (2): 420–425. doi:10.1378/chest.08-2427. PMID 19349380.
- ↑ "Provisional CDC Guidelines for the Use and Safety Monitoring of Bedaquiline Fumarate (Sirturo) for the Treatment of Multidrug-Resistant Tuberculosis".
- ↑ "WHO Disease and injury country estimates". World Health Organization. 2004. Retrieved 11 November 2009.
- ↑ Lambert M, et al. (2003). "Recurrence in tuberculosis: relapse or reinfection?". Lancet Infect Dis. 3 (5): 282–7. doi:10.1016/S1473-3099(03)00607-8. PMID 12726976.
- ↑ Wang, JY; Lee, LN; Lai, HC; Hsu, HL; Liaw, YS; Hsueh, PR; Yang, PC (15 July 2007). "Prediction of the tuberculosis reinfection proportion from the local incidence". The Journal of Infectious Diseases. 196 (2): 281–8. doi:10.1086/518898. PMID 17570116.
- ↑ World Health Organization (2009). "The Stop TB Strategy, case reports, treatment outcomes and estimates of TB burden". Global tuberculosis control: epidemiology, strategy, financing. pp. 187–300. ISBN 978-92-4-156380-2. Retrieved 14 November 2009.
- ↑ "Fact Sheets: The Difference Between Latent TB Infection and Active TB Disease". Centers for Disease Control. 20 June 2011. Retrieved 26 July 2011.
- ↑ "Global tuberculosis report 2013". World Health Organization. 2013.
- ↑ Lozano, R (15 December 2012). "Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010". Lancet. 380 (9859): 2095–128. doi:10.1016/S0140-6736(12)61728-0. PMID 23245604.
- 1 2 3 "Global Tuberculosis Control 2011" (PDF). World Health Organization. Retrieved 15 April 2012.
- ↑ World Health Organization (2009). "Epidemiology" (PDF). Global tuberculosis control: epidemiology, strategy, financing. pp. 6–33. ISBN 978-92-4-156380-2.
- ↑ FitzGerald, JM; Wang, L; Elwood, RK (8 February 2000). "Tuberculosis: 13. Control of the disease among aboriginal people in Canada". Canadian Medical Association Journal. 162 (3): 351–5. PMC 1231016
 . PMID 10693593.
. PMID 10693593. - ↑ Quah, Stella R.; Carrin, Guy; Buse, Kent; Kristian Heggenhougen (2009). Health Systems Policy, Finance, and Organization. Boston: Academic Press. p. 424. ISBN 0-12-375087-3.
- ↑ Anne-Emanuelle Birn (2009). Textbook of International Health: Global Health in a Dynamic World. p. 261. ISBN 9780199885213.
- ↑ Centers for Disease Control and Prevention. "CDC Surveillance Slides 2012 – TB".
- ↑ World Health Organization. "Global Tuberculosis Control Report, 2006 – Annex 1 Profiles of high-burden countries" (PDF). Retrieved 13 October 2006.
- ↑ Centers for Disease Control and Prevention (12 September 2006). "2005 Surveillance Slide Set". Retrieved 13 October 2006.
- ↑ Rothschild BM, Martin LD, Lev G, et al. (August 2001). "Mycobacterium tuberculosis complex DNA from an extinct bison dated 17,000 years before the present". Clin. Infect. Dis. 33 (3): 305–11. doi:10.1086/321886. PMID 11438894.
- ↑ Pearce-Duvet J (2006). "The origin of human pathogens: evaluating the role of agriculture and domestic animals in the evolution of human disease". Biol Rev Camb Philos Soc. 81 (3): 369–82. doi:10.1017/S1464793106007020. PMID 16672105.
- ↑ Comas, I; Gagneux, S (October 2009). Manchester, Marianne, ed. "The past and future of tuberculosis research". PLoS Pathogens. 5 (10): e1000600. doi:10.1371/journal.ppat.1000600. PMC 2745564
 . PMID 19855821.
. PMID 19855821. - ↑ Zink A, Sola C, Reischl U, Grabner W, Rastogi N, Wolf H, Nerlich A (2003). "Characterization of Mycobacterium tuberculosis Complex DNAs from Egyptian Mummies by Spoligotyping". J Clin Microbiol. 41 (1): 359–67. doi:10.1128/JCM.41.1.359-367.2003. PMC 149558
 . PMID 12517873.
. PMID 12517873. - ↑ Konomi N, Lebwohl E, Mowbray K, Tattersall I, Zhang D (2002). "Detection of Mycobacterial DNA in Andean Mummies". J Clin Microbiol. 40 (12): 4738–40. doi:10.1128/JCM.40.12.4738-4740.2002. PMC 154635
 . PMID 12454182.
. PMID 12454182. - ↑ Sledzik, Paul S.; Nicholas Bellantoni (June 1994). "Bioarcheological and biocultural evidence for the New England vampire folk belief" (PDF). American Journal of Physical Anthropology. 94 (2): 269–274. doi:10.1002/ajpa.1330940210. PMID 8085617.
- ↑ Léon Charles Albert Calmette at Who Named It?
- ↑ Trail RR (April 1970). "Richard Morton (1637–1698)". Med Hist. 14 (2): 166–74. doi:10.1017/S0025727300015350. PMC 1034037
 . PMID 4914685.
. PMID 4914685. - ↑ Zur Pathogenie der Impetigines. Auszug aus einer brieflichen Mitteilung an den Herausgeber. [Müller's] Archiv für Anatomie, Physiologie und wissenschaftliche Medicin. 1839, page 82.
- ↑ Kentucky: Mammoth Cave long on history. CNN. 27 February 2004. Accessed 8 October 2006.
- 1 2 3 McCarthy OR (August 2001). "The key to the sanatoria". J R Soc Med. 94 (8): 413–7. PMC 1281640
 . PMID 11461990.
. PMID 11461990. - ↑ Nobel Foundation. The Nobel Prize in Physiology or Medicine 1905. Accessed 7 October 2006.
- ↑ Waddington K (January 2004). "To stamp out "So Terrible a Malady": bovine tuberculosis and tuberculin testing in Britain, 1890–1939". Med Hist. 48 (1): 29–48. doi:10.1017/S0025727300007043. PMC 546294
 . PMID 14968644.
. PMID 14968644. - ↑ Bonah C (2005). "The 'experimental stable' of the BCG vaccine: safety, efficacy, proof, and standards, 1921–1933". Stud Hist Philos Biol Biomed Sci. 36 (4): 696–721. doi:10.1016/j.shpsc.2005.09.003. PMID 16337557.
- ↑ Comstock G (1994). "The International Tuberculosis Campaign: a pioneering venture in mass vaccination and research". Clin Infect Dis. 19 (3): 528–40. doi:10.1093/clinids/19.3.528. PMID 7811874.
- ↑ Bloom, editor, Barry R. (1994). Tuberculosis: pathogenesis, protection, and control. Washington, D.C.: ASM Press. ISBN 978-1-55581-072-6.
- ↑ Persson, Sheryl (2010). Smallpox, Syphilis and Salvation: Medical Breakthroughs That Changed the World. ReadHowYouWant.com. p. 141. ISBN 978-1-4587-6712-7.
- ↑ editor, Caroline Hannaway, (2008). Biomedicine in the twentieth century: practices, policies, and politics. Amsterdam: IOS Press. p. 233. ISBN 978-1-58603-832-8.
- ↑ Shields, Thomas (2009). General thoracic surgery (7th ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 792. ISBN 978-0-7817-7982-1.
- ↑ Lalloo UG, Naidoo R, Ambaram A (May 2006). "Recent advances in the medical and surgical treatment of multi-drug resistant tuberculosis". Curr Opin Pulm Med. 12 (3): 179–85. doi:10.1097/01.mcp.0000219266.27439.52. PMID 16582672.
- ↑ "Frequently asked questions about TB and HIV". World Health Organization. Retrieved 15 April 2012.
- ↑ "Hippocrates 3.16 Classics, MIT". Archived from the original on 11 February 2005. Retrieved 15 December 2015.
- ↑ Caldwell, Mark (1988). The Last Crusade. New York: Macmillan. p. 21. ISBN 0689118104.
- ↑ Bunyan, John. "The Life and Death of Mr. Badman". Google Books. Google. Retrieved 28 September 2016.
- 1 2 "Public-Private Partnership Announces Immediate 40 Percent Cost Reduction for Rapid TB Test" (pdf). World Health Organization. 6 August 2012.
- ↑ Lawn, SD; Nicol, MP (September 2011). "Xpert® MTB/RIF assay: development, evaluation and implementation of a new rapid molecular diagnostic for tuberculosis and rifampicin resistance". Future microbiology. 6 (9): 1067–82. doi:10.2217/fmb.11.84. PMC 3252681
 . PMID 21958145.
. PMID 21958145. - ↑ "WHO says Cepheid rapid test will transform TB care". Reuters. 8 December 2010.
- ↑ STOPTB (5 April 2013). "The Stop TB Partnership, which operates through a secretariat hosted by the World Health Organization (WHO) in Geneva, Switzerland." (pdf).
- ↑ Lienhardt, C; Espinal, M; Pai, M; Maher, D; Raviglione, MC (November 2011). "What research is needed to stop TB? Introducing the TB Research Movement". PLOS Medicine. 8 (11): e1001135. doi:10.1371/journal.pmed.1001135. PMC 3226454
 . PMID 22140369.
. PMID 22140369. - ↑ Mishra, G (2013). "Tuberculosis Prescription Practices In Private And Public Sector In India". NJIRM. 4 (2): 71–78.
- ↑ Anurag Bhargava; Lancelot Pinto; Madhukar Pai (2011). "Mismanagement of tuberculosis in India: Causes, consequences, and the way forward". Hypothesis. 9 (1): e7. doi:10.5779/hypothesis.v9i1.214.
- ↑ Amdekar, Y (July 2009). "Changes in the management of tuberculosis". Indian journal of pediatrics. 76 (7): 739–42. doi:10.1007/s12098-009-0164-4. PMID 19693453.
- 1 2 3 4 5 6 7 Courtwright, A; Turner, AN (Jul–Aug 2010). "Tuberculosis and stigmatization: pathways and interventions". Public Health Reports. 125 Suppl 4: 34–42. PMID 20626191.
- ↑ Mason, PH; Roy, A; Spillane, J; Singh, P (22 May 2015). "SOCIAL, HISTORICAL AND CULTURAL DIMENSIONS OF TUBERCULOSIS.". Journal of biosocial science: 1–27. doi:10.1017/S0021932015000115. PMID 25997539.
- 1 2 3 Martín Montañés, C.; Gicquel, B. (March 2011). "New tuberculosis vaccines". Enfermedades infecciosas y microbiologia clinica. 29 Suppl 1: 57–62. doi:10.1016/S0213-005X(11)70019-2. PMID 21420568.
- ↑ Ibanga H.; Brookes R.; Hill P.; Owiafe P.; Fletcher H.; Lienhardt C.; Hill A.; Adegbola R.; McShane H. (2006). "Early clinical trials with a new tuberculosis vaccine, MVA85A, in tuberculosis-endemic countries: issues in study design". Lancet Infectious Diseases. 6 (8): 522–8. doi:10.1016/S1473-3099(06)70552-7. PMID 16870530.
- ↑ Kaufmann S.H. (2010). "Future vaccination strategies against tuberculosis: Thinking outside the box". Immunity. 33 (4): 567–77. doi:10.1016/j.immuni.2010.09.015. PMID 21029966.
- ↑ Webber D.; Kremer M. (2001). "Stimulating Industrial R&D for Neglected Infectious Diseases: Economic Perspectives" (PDF). Bulletin of the World Health Organization. 79 (8): 693–801.
- ↑ Barder O.; Kremer M.; Williams H. (2006). "Advance Market Commitments: A Policy to Stimulate Investment in Vaccines for Neglected Diseases". The Economists' Voice. 3 (3). doi:10.2202/1553-3832.1144.
- ↑ Economic, Department of; Affairs, Social (2009). Achieving the global public health agenda: dialogues at the Economic and Social Council. New York: United Nations. p. 103. ISBN 978-92-1-104596-3.
- ↑ Jong, [edited by] Jane N. Zuckerman, Elaine C. (2010). Travelers' vaccines (2nd ed.). Shelton, CT: People's Medical Publishing House. p. 319. ISBN 978-1-60795-045-5.
- ↑ Bill and Melinda Gates Foundation Announcement (12 February 2004). "Gates Foundation Commits $82.9 Million to Develop New Tuberculosis Vaccines".
- ↑ Nightingale, Katherine (19 September 2007). "Gates foundation gives US$280 million to fight TB".
- 1 2 Zumla, A; Hafner, R; Lienhardt, C; Hoelscher, M; Nunn, A (1 March 2012). "Advancing the development of tuberculosis therapy". Nature reviews. Drug discovery. 11 (3): 171–2. doi:10.1038/nrd3694. PMID 22378254.
- ↑ "J&J Sirturo Wins FDA Approval to Treat Drug-Resistant TB". Bloombeg. 31 December 2012. Retrieved 1 January 2013.
- 1 2 Avorn, J (April 2013). "Approval of a tuberculosis drug based on a paradoxical surrogate measure". JAMA. 309 (13): 1349–1350. doi:10.1001/jama.2013.623. PMID 23430122.
- ↑ US Food and Drug Administration. "Briefing Package: NDA 204–384: Sirturo" (PDF).
- ↑ Zuckerman, Diana; Jennifer Yttri (January 2013). "Antibiotics: When science and wishful thinking collide". Health Affairs.
- ↑ Shivaprasad, H.L.; Palmieri, C. (January 2012). "Pathology of mycobacteriosis in birds". The Veterinary Clinics of North America. Exotic Animal Practice. 15 (1): 41–55, v–vi. doi:10.1016/j.cvex.2011.11.004. PMID 22244112.
- ↑ Reavill, D.R.; Schmidt, R.E. (January 2012). "Mycobacterial lesions in fish, amphibians, reptiles, rodents, lagomorphs, and ferrets with reference to animal models". The Veterinary Clinics of North America. Exotic Animal Practice. 15 (1): 25–40, v. doi:10.1016/j.cvex.2011.10.001. PMID 22244111.
- ↑ Mitchell, M.A. (January 2012). "Mycobacterial infections in reptiles". The Veterinary Clinics of North America. Exotic Animal Practice. 15 (1): 101–11, vii. doi:10.1016/j.cvex.2011.10.002. PMID 22244116.
- ↑ Wobeser, Gary A. (2006). Essentials of disease in wild animals (1st ed.). Ames, Iowa [u.a.]: Blackwell Publishing. p. 170. ISBN 978-0-8138-0589-4.
- ↑ Ryan, T.J.; Livingstone, P.G.; Ramsey, D.S.; de Lisle, G.W.; Nugent, G.; Collins, D.M.; Buddle, B.M. (25 February 2006). "Advances in understanding disease epidemiology and implications for control and eradication of tuberculosis in livestock: the experience from New Zealand". Veterinary Microbiology. 112 (2–4): 211–9. doi:10.1016/j.vetmic.2005.11.025. PMID 16330161.
- ↑ White, P.C.; Böhm, M.; Marion, G.; Hutchings, M.R. (September 2008). "Control of bovine tuberculosis in British livestock: there is no 'silver bullet'". Trends in Microbiology. 16 (9): 420–7. doi:10.1016/j.tim.2008.06.005. PMID 18706814.
- ↑ Ward, A.I.; Judge, J.; Delahay, R.J. (1 January 2010). "Farm husbandry and badger behaviour: opportunities to manage badger to cattle transmission of Mycobacterium bovis?". Preventive veterinary medicine. 93 (1): 2–10. doi:10.1016/j.prevetmed.2009.09.014. PMID 19846226.
- ↑ Holt, Nathalia (24 March 2015). "The Infected Elephant in the Room". Slate. Retrieved 2016-04-05.
- ↑ Mikota, Susan K. "A Brief History of TB in Elephants" (PDF). APHIS. US Department of Agriculture. Retrieved 2016-04-05.
External links
| Wikiquote has quotations related to: Tuberculosis |
| Wikimedia Commons has media related to Tuberculosis. |
- Tuberculosis at DMOZ
- "Tuberculosis (TB)". Centers for Disease Control.
- "Tuberculosis (TB)". UK Health Protection Agency.
