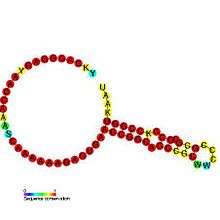
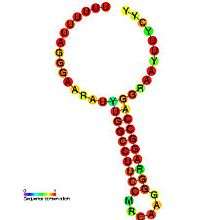
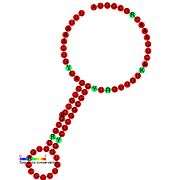
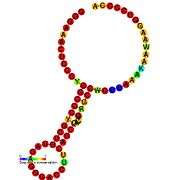
Translational frameshift
Translational frameshifting or ribosomal frameshifting refers to an alternative process of protein translation. A protein is translated from one end of the mRNA to the other, from the 5' to the 3' end. Normally a protein is translated from a template mRNA with consecutive blocks of 3 nucleotides being read as single amino acids. However, certain organisms may exhibit a change or shift in the ribosomes frame by one or two nucleotides when translating the genetic code. This is deemed translational or ribosomal frameshifting.[1] The process can be programmed by the nucleotide sequence of the mRNA and is sometimes affected by the secondary or tertiary mRNA structure. It has been described mainly in viruses (especially retroviruses), retrotransposons and bacterial insertion elements, and also in some cellular genes.
Process overview
Proteins are translated unidirectionally by reading tri-nucleotides on the mRNA strand also known as codons. Therefore, a shift of any number of nucleotides that is not divisible by 3 in the reading frame will result in subsequent codons to be read differently.[2] This effectively changes the ribosomal reading frame. For example, the following sentence when read from the beginning makes sense to a reader:
|Start|THE CAT AND THE MAN ARE FAT ... |Start|123 123 123 123 123 123 123 ...
However, changing the reading frame by say shifting the first reading up one letter between the T and H on the first word:
T|Start|HEC ATA NDT HEM ANA REF AT... -|Start|123 123 123 123 123 123 12...
Now the sentence makes absolutely no sense. In the case of a translating ribosome, a frameshift can result in nonsense being created after the frameshift or a completely different protein being created after the frameshift. When referring to translational frameshifting, the latter is always implied, the former being usually a result of a point mutation such as a deletion.
Controlling mechanisms
The main distinction between frameshifts resulting from mutation and those resulting from ribosomal frameshifting is that the latter are controlled by various mechanisms found in codons. These mechanisms emerge from the fact that ribosomes do not translate proteins at a steady rate, regardless of the sequence. Certain codons take longer to translate, because there are not equal amounts of tRNA of that particular codon in the cytosol.[3] Due to this lag, there exist in small sections of codons sequences that control the rate of ribosomal frameshifting. Sections of less accessible codons that slow ribosomal transaction are known as "choke points," and sections of easily accessible codons which result in faster ribosomal transaction are "slippery sequences." Slippery sequences can potentially make the reading ribosome "slip" and skip a number of nucleotides (usually only 1) and read a completely different frame hereafter. Choke points reduce the probability of this happening.[4]
Examples
This type of frameshifting may be programmed to occur at particular recoding sites and is important in some viruses (e.g. SARS, HIV[5]) and some cellular genes (e.g. prfB a release factor). Its use is primarily for compacting more genetic information into a shorter amount of genetic material.
See also
- Antizyme RNA frameshifting stimulation element
- Coronavirus frameshifting stimulation element
- DnaX ribosomal frameshifting element
- Frameshift mutation
- HIV ribosomal frameshift signal
- Insertion sequence IS1222 ribosomal frameshifting element
- Recode database
- Ribosomal frameshift
- Ribosomal pause
- Slippery sequence
References
- ↑ Léger M, Dulude D, Steinberg SV, Brakier-Gingras L (2007). "The three transfer RNAs occupying the A, P and E sites on the ribosome are involved in viral programmed -1 ribosomal frameshift". Nucleic Acids Res. 35 (16): 5581–92. doi:10.1093/nar/gkm578. PMC 2018615
 . PMID 17704133.
. PMID 17704133. - ↑ Ivanov IP, Atkins JF (2007). "Ribosomal frameshifting in decoding antizyme mRNAs from yeast and protists to humans: close to 300 cases reveal remarkable diversity despite underlying conservation". Nucleic Acids Res. 35 (6): 1842–58. doi:10.1093/nar/gkm035. PMC 1874602
 . PMID 17332016.
. PMID 17332016. - ↑ de Crécy-Lagard V (2007). "Identification of genes encoding tRNA modification enzymes by comparative genomics". Meth. Enzymol. 425: 153–83. doi:10.1016/S0076-6879(07)25007-4. PMC 3034448
 . PMID 17673083.
. PMID 17673083. - ↑ Green L, Kim CH, Bustamante C, Tinoco I (January 2008). "Characterization of the mechanical unfolding of RNA pseudoknots". J. Mol. Biol. 375 (2): 511–28. doi:10.1016/j.jmb.2007.05.058. PMID 18021801.
- ↑ McNaughton BR, Gareiss PC, Miller BL (September 2007). "Identification of a selective small-molecule ligand for HIV-1 frameshift-inducing stem-loop RNA from an 11,325 member resin bound dynamic combinatorial library". J. Am. Chem. Soc. 129 (37): 11306–7. doi:10.1021/ja072114h. PMID 17722919.
- ↑ Mazauric, H.; Licznar, P.; Prère, F.; Canal, I.; Fayet, O. (July 2008). "Apical loop-internal loop RNA pseudoknots: a new type of stimulator of -1 translational frameshifting in bacteria". J Biol Chem. 283 (29): 20421–32. doi:10.1074/jbc.M802829200. PMID 18474594.
- ↑ Ivanov IP, Anderson CB, Gesteland RF, Atkins JF (2004). "Identification of a new antizyme mRNA +1 frameshifting stimulatory pseudoknot in a subset of diverse invertebrates and its apparent absence in intermediate species.". J Mol Biol. 339 (3): 495–504. doi:10.1016/j.jmb.2004.03.082. PMID 15147837.
- ↑ Baranov, PV; Henderson CM; Anderson CB; Gesteland RF; Atkins JF; Howard MT (2005). "Programmed ribosomal frameshifting in decoding the SARS-CoV genome". Virology. 332 (2): 498–510. doi:10.1016/j.virol.2004.11.038. PMID 15680415.
- ↑ Larsen, B; Gesteland RF; Atkins JF (1997). "Structural probing and mutagenic analysis of the stem-loop required for Escherichia coli dnaX ribosomal frameshifting: programmed efficiency of 50%.". J Mol Biol. 271 (1): 47–60. doi:10.1006/jmbi.1997.1162. PMID 9300054.
External links
- Frameshifting, Ribosomal at the US National Library of Medicine Medical Subject Headings (MeSH)
- Wise2 — aligns a protein against a DNA sequence allowing frameshifts and introns
- FastY — compare a DNA sequence to a protein sequence database, allowing gaps and frameshifts
- Path — tool that compares two frameshift proteins (back-translation principle)
- Recode2 — Database of recoded genes, including those that require programmed Translational frameshift.