Species complex
In biology, a species complex is a group of closely related species that are very similar in appearance to the point that the boundaries between them are often unclear. Terms sometimes used synonymously but with more precise meanings are: cryptic species for two or more species hidden under one species name, sibling species for two cryptic species that are each other's closest relative, and species flock for a group of closely related species living in the same habitat. As informal taxonomic ranks, species group, species aggregate, and superspecies are also in use.
Two or more taxa once considered conspecific (of the same species) may later be subdivided into infraspecific taxa (taxa within a species, such as bacterial strains or plant varieties), but this is not a species complex.
A species complex is in most cases a monophyletic group with a common ancestor, although there are exceptions. It may represent an early stage after speciation, but may also have been separated for a long time period without evolving morphological differences. Hybrid speciation can be a component in the evolution of a species complex.
Species complexes exist in all groups of organisms. They are identified by the rigorous study of differences between individual species, making use of minute morphological details, tests of reproductive isolation, or DNA-based methods such as molecular phylogenetics or DNA barcoding. The existence of extremely similar species may cause local and global species diversity to be underestimated. Recognizing similar but distinct species is important for disease and pest control, and in conservation biology, although drawing dividing lines between species can be inherently difficult.
Definition
A species complex is typically considered as a group of close, but distinct species.[5] Obviously, the concept is closely tied to the definition of a species. Modern biology understands a species as "separately evolving metapopulation lineage" but acknowledges that the criteria to delimit species may depend on the group studied.[6] Thus, many species defined traditionally, based only on morphological similarity, have been found to comprise several distinct species when other criteria, such as genetic differentiation or reproductive isolation were applied.[7]
A more restricted use applies the term to close species between which hybridisation occurred or is occurring, leading to intermediate forms and blurred species boundaries.[8]
Some authors apply the term also to a species with intraspecific variability, which might be a sign of ongoing or incipient speciation. Examples are ring species[9][10] or species with subspecies, where it is often unclear if these should be considered separate species.[11]
Included concepts
Several terms are used synonymously for a species complex, but some of them may also have slightly different, or more narrow meanings. In the nomenclature codes of zoology and bacteriology, no taxonomic ranks are defined at the level between subgenera and species,[12][13] while the botanical code defines four ranks below genera (section, subsections, series and subseries).[14] Different informal taxonomic solutions have been used to indicate a species complex.
- Cryptic species
- Also physiologic race[15] (uncommon). This describes "distinct species that are erroneously classified (and hidden) under one species name".[16] More generally, the term is often applied when species, even if known to be distinct, cannot be reliably distinguished based on their morphology. The usage physiologic race is not to be confused with physiological race.
- Sibling species
- Also aphanic species. This term, introduced by Ernst Mayr in 1942,[17] was initially used with the same meaning as cryptic species,[7] but later authors emphasized the common phylogenetic origin.[18] A recent article defines sibling species as "cryptic sister species", meaning "two species that are the closest relative of each other and have not been distinguished from one another taxonomically".[16]
- Species flock
- Also species swarm. This refers to "a monophyletic group of closely related species all living in the same ecosystem".[16] Conversely, the term has also been applied very broadly to a group of closely related species than can be variable and widespread.[19]
- Superspecies
- Sometimes used as an informal rank for a species complex around one "representative" species.[20][21] Popularized by Bernhard Rensch and later Ernst Mayr, with the initial requirement that species forming a superspecies must have allopatric distributions.[22] For the component species of a superspecies, allospecies was proposed.[22]
- Species aggregate
- Used for a species complex, especially in plant taxa where polypoidy and apomixis are common. Historical synonyms are species collectiva, introduced by Adolf Engler, conspecies, and grex.[23] Components of a species aggregate have been called segregates or microspecies.[23] Used as abbreviation agg. after the binomial species name.[8][24]
- Sensu lato
- Meaning "in the broad sense", this Latin phrase is often used after a binomial species name, often abbreviated as s.l., to indicate a species complex represented by that species.[25][26][27]
Identification
Distinguishing close species within a complex requires the study of often very small differences. Morphological differences may be minute and only visible using adapted methods, such as microscopy. However, distinct species may sometimes have no morphological differences.[16] In these cases, other characters, e.g. in the species' life history, behavior, physiology, or karyology can be explored. As an example, territorial songs are indicative of species in the treecreepers, a bird genus with little morphological differences.[28] Mating tests are common in some groups such as fungi to confirm the reproductive isolation of two species.[26]
Analysis of DNA sequences is becoming increasingly standard for species recognition and may in many cases be the only useful method.[16] Different methods are used to analyse such genetic data, for example molecular phylogenetics or DNA barcoding. Such methods have greatly contributed to the discovery of cryptic species,[16] including such emblematic species as the fly agaric[2] or the African elephants.[3]
Evolution and ecology
Speciation process
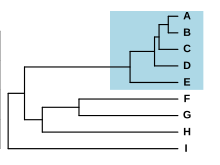
Species forming a complex have typically diverged very recently from each other, allowing in some cases to retrace the process of speciation. Species with differentiated populations such as ring species are sometimes seen as an example of early, ongoing speciation, i.e. a species complex in formation. Nevertheless, similar but distinct species have sometimes been isolated for a long time without evolving differences, a phenomenon called "morphological stasis".[16] As an examples, the Amazonian frog Pristimantis ockendeni is actually at least three different species that diverged over 5 million years ago.[30]
Stabilizing selection has been invoked as a force maintaining similarity in species complexes, especially when adaptation to special environments, such as a host in the case of symbionts, or extreme environments, constrains possible directions of evolution: In such cases, strongly divergent selection is not to be expected.[16] Also, asexual reproduction, such as through apomixis in plants, may separate lineages without producing a great degree of morphological differentiation.
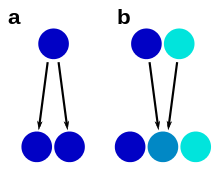
a – morphological stasis
b – hybrid speciation
A species complex is usually a group that has one common ancestor (a monophyletic group), although closer examination can sometimes disprove this. As an example, the yellow-spotted "fire salamanders" in the genus Salamandra, formerly all classified as one species S. salamandra, are not monophyletic: the Corsican fire salamander's closest relative was shown to be the entirely black Alpine salamander.[29] In such cases, similarity has arisen from convergent evolution.
Hybrid speciation can lead to unclear species boundaries through a process of reticulate evolution, where species have two parent species as their most recent common ancestors. In such cases, the hybrid species may have intermediate characters, as demonstrated e.g. in Heliconius butterflies.[31] Hybrid speciation has been observed in various species complexes, such as insects, fungi, and plants. In plants, hybridization often takes place through polyploidization, and hybrid plant species are called nothospecies.
Range
In regards to whether or not members of a species group share a range, sources differ. A source from Iowa State University Department of Agronomy says that members of a species group usually have partially overlapping ranges but do not interbreed with each other.[32] A Dictionary of Zoology (Oxford University Press 1999) describes a species group as complex of related species that exist allopatrically and explains that this "grouping can often be supported by experimental crosses in which only certain pairs of species will produce hybrids."[33] The examples given below may support both uses of the term "species group."
Often such complexes only become evident when a new species is introduced into the system, breaking down existing species barriers. An example is the introduction of the Spanish slug in Northern Europe, where interbreeding with the local black slug and red slug, traditionally considered clearly separate species that did not interbreed, shows they may be actually just subspecies of the same species.[34]
Where closely related species coexist in sympatry, it is often a particular challenge to understand how these similar species persist without outcompeting each other. Niche partitioning is one mechanism invoked to explain this. Studies in some species complexes indeed suggest that species divergence went in par with ecological differentiation, with species now preferring different microhabitats. Similar methods also found that the Amazonian frog Eleutherodactylus ockendeni is actually at least 3 different species that diverged over 5 million years ago.[30] A species flock may arise when a species penetrates a new geographical area and diversifies to occupy a variety of ecological niches; this process is known as adaptive radiation. The first species flock to be recognized as such was the 13 species of Darwin's finches on the Galápagos Islands described by Charles Darwin.
Practical implications
Biodiversity estimates
It has been suggested that cryptic species complexes are very common in the marine environment.[35] Although this suggestion came before the detailed analysis of many systems using DNA sequence data, it has been proven correct.[36] The increased use of DNA sequence in the investigation of organismal diversity (also called Phylogeography and DNA barcoding) has led to the discovery of a great many cryptic species complexes in all habitats. In the marine bryozoan Celleporella hyalina,[37] detailed morphological analyses and mating compatibility tests between the isolates identified by DNA sequence analysis were used to confirm that these groups consisted of more than 10 ecologically distinct species that had been diverging for many million years.
Evidence from the identification of cryptic species has led some to conclude that current estimates of global species richness are too low.
Disease and pathogen control

Pests, species causing diseases, and their vectors, have direct importance for humans. When they are found to be cryptic species complexes, the ecology and virulence of each of these species needs to be reevaluated to devise appropriate control strategies. An example are cryptic species in the malaria vector Anopheles, or the fungi causing cryptococcosis.
Conservation biology
When a species is found to comprise in fact several phylogenetically distinct species, each of these typically have smaller distribution ranges and population sizes than reckoned before. These different species can also differ in their ecology, e.g. having different breeding strategies or habitat requirements, which has to be taken into account for appropriate management. For example, giraffe populations and subspecies differ genetically to such an extent that they may be considered species; while the giraffe as a whole is not considered threatened, considering each cryptic species separately would mean a much higher level of threat.[39]
See also
References
- ↑ Ron, Santiago; Caminer, Marcel (2014). "Systematics of treefrogs of the Hypsiboas calcaratus and Hypsiboas fasciatus species complex (Anura, Hylidae) with the description of four new species". ZooKeys. 370: 1–68. doi:10.3897/zookeys.370.6291. ISSN 1313-2970.
- 1 2 Geml J, Tulloss RE, Laursen GA, Sasanova NA, Taylor DL (2008). "Evidence for strong inter- and intracontinental phylogeographic structure in Amanita muscaria, a wind-dispersed ectomycorrhizal basidiomycete" (PDF). Molecular Phylogenetics and Evolution. 48 (2): 694–701. doi:10.1016/j.ympev.2008.04.029. PMID 18547823.
- 1 2 Roca AL, Georgiadis N, Pecon-Slattery J, O'Brien SJ (2001). "Genetic evidence for two species of elephant in Africa". Science. 293 (5534): 1473–1477. doi:10.1126/science.1059936. PMID 11520983.
- ↑ Moran P, Kornfield I (1993). "Retention of an Ancestral Polymorphism in the Mbuna Species Flock (Teleostei: Cichlidae) of Lake Malawi" (PDF). Molecular Biology and Evolution. 10 (5): 1015–1029.
- ↑ Brown JK, Frohlich DR, Rosell RC (1995). "The sweetpotato or silverleaf whiteflies: biotypes of Bemisia tabaci or a species complex?". Annual Review of Entomology. 40 (1): 511–534. doi:10.1146/annurev.en.40.010195.002455.
- ↑ De Queiroz, Kevin (2007). "Species Concepts and Species Delimitation". Systematic Biology. 56 (6): 879–886. doi:10.1080/10635150701701083. ISSN 1063-5157.
- 1 2 Mayr E. (1970). "Morphological species characters and sibling species". Populations, Species, and Evolution. Cambridge, MA: The Belknap Press of Harvard University Press. pp. 21–36. ISBN 0-674-69013-3.
- 1 2 Horandl, E.; Greilhuber, J.; Klimova, K.; Paun, O.; Temsch, E.; Emadzade, K.; Hodalova, I. (2009). "Reticulate evolution and taxonomic concepts in the Ranunculus auricomus complex (Ranunculaceae): insights from analysis of morphological, karyological and molecular data". Taxon. 58 (4): 1194–1215. PMC 2855680
 . PMID 20401184.
. PMID 20401184. - ↑ Moritz C, Schneider CJ, Wake DB (1992). "Evolutionary relationships within the Ensatina eschscholtzii complex confirm the ring species interpretation". Systematic Biology. 41 (3): 273–291. doi:10.1093/sysbio/41.3.273.
- ↑ Bowen BW, Bass AL, Rocha LA, Grant WS, Roberston DR (2001). "Phylogeography of the trumpetfishes (Aulostomus): Ring species complex on a global scale". Evolution. 55 (5): 1029–1039. doi:10.1111/j.0014-3820.2001.tb00619.x.
- ↑ Muñoz MM; Crawford NG; McGreevy Jr. TJ; Messana NJ; Tarvin RD; Revell LJ; Zandvliet RM; Hopwood JM; Mock E; Schneider AL; Schneider CJ. (2013). "Divergence in coloration and ecological speciation in the Anolis marmoratus species complex" (PDF). Molecular Ecology. 22 (10): 2668–2682. doi:10.1111/mec.12295. PMID 23611648.
- ↑ Ride WD, Cogger HG, Dupuis C, Kraus O, Minelli A, Thompson FC, Tubbs PK, eds. (1999). "Chapter 9: Genus-group nominal taxa and their names". International Code of Zoological Nomenclature. London: The International Trust for Zoological Nomenclature. ISBN 0853010064.
- ↑ Lapage SP, Sneath PH, Lessel EF, Skerman VB, Seeliger HP, Clark WA, eds. (1992). "Chapter 3. Rules of nomenclature with recommendations. Section 2. Ranks of taxa". International Code of Nomenclature of Bacteria. Bacteriological Code, 1990 Revision. Washington (DC): ASM Press.
- ↑ McNeill, J; et al., eds. (2012), International Code of Nomenclature for algae, fungi, and plants (Melbourne Code), adopted by the Eighteenth International Botanical Congress Melbourne, Australia, July 2011 (electronic ed.), Bratislava: International Association for Plant Taxonomy, retrieved 2012-12-20.
- ↑ Morris, Christopher, ed. (1992). "Physiologic race". Academic Press Dictionary of Science and Technology. San Diego / London: Academic Press. p. 1643. ISBN 0-12-200400-0.
- 1 2 3 4 5 6 7 8 Bickford D, Lohman DJ, Sodhi NS, Ng PK, Meier R, Winker K, Ingram KK, Das I (2007). "Cryptic species as a window on diversity and conservation" (PDF). Trends in Ecology & Evolution. 22 (3): 148–155. doi:10.1016/j.tree.2006.11.004.
- ↑ Mayr, Ernst (1942). Systematics and the Origin of Species, from the Viewpoint of a Zoologist. ISBN 9780674862500.
- ↑ Steyskal GC. (1972). "The meaning of the term 'sibling species'" (PDF). Systematic Zoology. 21 (4): 446. doi:10.1093/sysbio/21.4.446.
- ↑ Hodges SA, Arnold ML (1994). "Columbines: a geographically widespread species flock" (PDF). Proceedings of the National Academy of Sciences of the United States of America. 91 (11): 5129–5132. doi:10.1073/pnas.91.11.5129. PMC 43945
 . PMID 8197196.
. PMID 8197196. - ↑ Fontdevila A, Pla C, Hasson E, Wasserman M, Sanchez A, Naveira H, Ruiz A (1988). "Drosophila koepferae: a new member of the Drosophila serido (Diptera: Drosophilidae) superspecies taxon". Annals of the Entomological Society of America. 81 (3): 380–385. doi:10.1093/aesa/81.3.380.
- ↑ Wallis GP, Arntzen JW (1989). "Mitochondrial-DNA variation in the crested newt superspecies: Limited cytoplasmic gene flow among species". Evolution. 43 (1): 88–104. doi:10.2307/2409166. JSTOR 2409166.
- 1 2 Amadon D. (1966). "The superspecies concept". Systematic Biology. 15 (3): 245–249. doi:10.2307/sysbio/15.3.245.
- 1 2 Heywood VH. (1962). "The "species aggregate" in theory and practice". In VH Heywood; Löve Á. Symposium on Biosystematics, organized by the International Organization of Biosystematists, Montreal, October 1962. pp. 26–36.
- ↑ Kankare M, Van Nouhuys S, Hanski I (2005). "Genetic divergence among host-specific cryptic species in Cotesia melitaearum aggregate (Hymenoptera: Braconidae), parasitoids of checkerspot butterflies". Annals of the Entomological Society of America. 98 (3): 382–394. doi:10.1603/0013-8746(2005)098[0382:GDAHCS]2.0.CO;2.
- ↑ Wallis GP, Judge KF, Bland J, Waters JM, Berra TM (2001). "Genetic diversity in New Zealand Galaxias vulgaris sensu lato (Teleostei: Osmeriformes: Galaxiidae): a test of a biogeographic hypothesis". Journal of Biogeography. 28 (1): 59–67. doi:10.1046/j.1365-2699.2001.00535.x.
- 1 2 Dai Y-C, Vainio EJ, Hantula J, Niemelä, Korhonen K (2003). "Investigations on Heterobasidion annosum s.lat. in central and eastern Asia with the aid of mating tests and DNA fingerprinting". Forest Pathology. 33 (5): 269–286. doi:10.1046/j.1439-0329.2003.00328.x.
- ↑ Van de Putte K, Nuytinck J, Stubbe D, Le HT, Verbeken A (2010). "Lactarius volemus sensu lato (Russulales) from northern Thailand: Morphological and phylogenetic species concepts explored". Fungal Diversity. 45 (1): 99–130. doi:10.1007/s13225-010-0070-0.
- ↑ Tietze DT, Martens J, Sun YH (2006). "Molecular phylogeny of treecreepers (Certhia) detects hidden diversity". Ibis. 148 (3): 477–488. doi:10.1111/j.1474-919X.2006.00547.x.

- 1 2 Steinfartz S, Veith M, Tautz D (2000). "Mitochondrial sequence analysis of Salamandra taxa suggests old splits of major lineages and postglacial recolonizations of Central Europe from distinct source populations of Salamandra salamandra". Molecular Ecology. 9 (4): 397–410. doi:10.1046/j.1365-294x.2000.00870.x. PMID 10736043.
- 1 2 Elmer K, Davila J, Lougheed S (2007). "Cryptic diversity and deep divergence in an upper Amazonian leaflitter frog, Eleutherodactylus ockendeni". BMC Evolutionary Biology. 7 (1): 247. doi:10.1186/1471-2148-7-247. ISSN 1471-2148. PMC 2254618
 . PMID 18154647.
. PMID 18154647. - ↑ Mallet J, Beltrán M, Neukirchen W, Linares M (2007). "Natural hybridization in heliconiine butterflies: the species boundary as a continuum". BMC Evolutionary Biology. 7 (1): 28. doi:10.1186/1471-2148-7-28. PMC 1821009
 . PMID 17319954.
. PMID 17319954. 
- ↑ Iowa State University Department of Agronomy
- ↑ Michael Allaby. "species group." A Dictionary of Zoology (Oxford University Press 1999)
- ↑ (Danish) Engelke, S. (2006?): Til Snegleforeningen (Note to the Danish Slug-society). Article in Danish
- ↑ Knowlton N (1993). "Sibling species in the sea". Annual Review of Ecology and Systematics. 24 (1): 189–216. doi:10.1146/annurev.es.24.110193.001201. ISSN 0066-4162.
- ↑ Knowlton N. (February 2000). "Molecular genetic analyses of species boundaries in the sea". Hydrobiologia. 420 (1): 73–90. doi:10.1023/A:1003933603879. ISSN 0018-8158.
- ↑ Gómez A, Wright PJ, Lunt DH, Cancino JM, Carvalho GR, Hughes RN (2007). "Mating trials validate the use of DNA barcoding to reveal cryptic speciation of a marine bryozoan taxon". Proceedings of the Royal Society B. 274 (1607): 199–207. doi:10.1098/rspb.2006.3718. ISSN 0962-8452. PMC 1685843
 . PMID 17035167.
. PMID 17035167. - ↑ Besansky NJ, Powell JR, Caccone A, Hamm DM, Scott JA, Collins FH (1994). "Molecular phylogeny of the Anopheles gambiae complex suggests genetic introgression between principal malaria vectors". Proceedings of the National Academy of Sciences of the United States of America. 91 (15): 6885–8. doi:10.1073/pnas.91.15.6885. PMC 44302
 . PMID 8041714.
. PMID 8041714. - ↑ Brown D, Brenneman R, Koepfli KP, Pollinger J, Mila B, Georgiadis N, Louis E, Grether G, Jacobs D, Wayne R (2007). "Extensive population genetic structure in the giraffe". BMC Biology. 5 (1): 57. doi:10.1186/1741-7007-5-57. ISSN 1741-7007. PMC 2254591
 . PMID 18154651.
. PMID 18154651.






.jpg)