Trimethylhexamethylenediamine
 | |
| Names | |
|---|---|
| IUPAC name
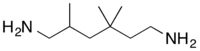
2,2,4-Trimethyl-1,6-hexanediamine | |
| Identifiers | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 69172 |
| |
| |
| Properties | |
| C9H22N2 | |
| Molar mass | 158.29 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
 | |
| Names | |
|---|---|
| IUPAC name
2,4,4-Trimethyl-1,6-hexanediamine | |
| Identifiers | |
| 3D model (Jmol) | Interactive image |
| ChemSpider | 83906 |
| |
| |
| Properties | |
| C9H22N2 | |
| Molar mass | 158.29 g·mol−1 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
trimethylhexamethylenediamine is the name used to refer to a mixture of two isomers of trimethyl-1,6-hexanediamine. The mixture is used as a monomer in nylon TMDT.
Trimethylhexamethylenediamine is synthesized from isophorone. Isophorone is reduced by hydrogenation to the trimethylcyclohexanol, which is then oxidized with nitric acid (in the same fashion as adipic acid is synthesized from cyclohexane). The diacid is converted to the diamine via the dinitrile.[2]
References
- 1 2 "TRIMETHYLHEXAMETHYLENEDIAMINE". chemicalbook.com. Retrieved 19 April 2015.
- ↑ U. Rohde-Liebenau (1995). "13.10 PA-TMDT". In Kohan, Melvin. Nylon Plastics Handbook. Munich: Hanser. p. 570. ISBN 1569901899.
This article is issued from Wikipedia - version of the 11/16/2015. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.