Microsatellite instability

Microsatellite instability (MSI) is the condition of genetic hypermutability that results from impaired DNA mismatch repair (MMR). The presence of MSI represents phenotypic evidence that MMR is not functioning normally.
MMR corrects errors that spontaneously occur during DNA replication, such as single base mismatches or short insertions and deletions. The proteins involved in MMR correct polymerase errors by forming a complex that binds to the mismatched section of DNA, excises the error, and inserts the correct sequence in its place.[1] Cells with abnormally functioning MMR are unable to correct errors that occur during DNA replication and consequently accumulate errors. This causes the creation of novel microsatellite fragments. Polymerase chain reaction-based assays can reveal these novel microsatellites and provide evidence for the presence of MSI.
Microsatellites are repeated sequences of DNA. These sequences can be made of repeating units of one to six base pairs in length. Although the length of these microsatellites is highly variable from person to person and contributes to the individual DNA "fingerprint", each individual has microsatellites of a set length. The most common microsatellite in humans is a dinucleotide repeat of the nucleotides C and A, which occurs tens of thousands of times across the genome. Microsatellites are also known as simple sequence repeats (SSRs).
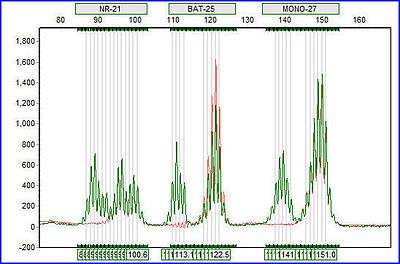
Structure
Microsatellite instability structure consists of repeated nucleotides, most often seen as GT/CA repeats.[2]
Researchers have yet to confirm the precise definition of the MSI structure. While all researchers agree that microsatellites are repeat sequences, the lengths of the sequences remain in question. Some research suggest that MSIs are short tandem DNA repeat sequences of one to six base pairs throughout the genome, while other research suggests that the range may be two to five.[3]
Although researchers do not agree on a specific threshold for the number of tandem repeats that constitute a microsatellite, there is a consensus around their relative size. Longer sequences are called minisatellite, and even longer sequences are called satellite DNA sites. Some scientists distinguish among the three categories by a minimum number of base pairs, and others use a minimum number of repeated units. The majority of repeats occur in untranslated regions, specifically introns. However, microsatellites that occur in coding regions often inhibit the expansion of most downstream events. Microsatellites make up approximately three percent of the human genome, or more than one million fragments of DNA. Microsatellite density increases with genome size and is seen twice as much at the ends of chromosome arms than in the chromosome bodies.[4]
Form and function
MSI was discovered in the 1970s and 1980s. The first human disease attributed to MSI was xeroderma pigmentosum. This disease resulted from two alleles activating mutations on nucleotide excision repair.
In a broad sense, MSI results from the inability of the mismatch repair (MMR) proteins to fix a DNA replication error. DNA replication occurs in the "S" phase of the cell cycle; the faulty event creating an MSI region occurs during the second replication event. The original strand is unharmed, but the daughter strand experiences a frame-shift mutation due to DNA polymerase slippage. Specifically, DNA polymerase slips, creating a temporary insertion-deletion loop, which is usually recognized by MMR proteins. However, when the MMR proteins do not function normally, as in the case of MSI, this loop results in frame-shift mutations, either through insertions or deletions, yielding non-functioning proteins.[5]
MSI is unique to DNA polymorphisms in that the replication errors vary in length instead of sequence. The rate and direction of the mutations yielding MSIs are the major components in determining genetic differences. To date, scientists agree that the mutation rates differ in loci position. The greater the length of the MSI, the greater the mutation rate.[4]
Although most mutations of MSI are the result of frame-shift mutations, occasionally the mutation events leading to MSI are derived from the hypermethylation of the hMLH1 (MMR protein) promoter. Hypermethylation occurs when a methyl group is added to a DNA nucleotide, resulting in gene silencing, thus yielding MSI.[6]
Researchers have shown that oxidative damage yields frame-shift mutations, thus yielding MSI, but they have yet to agree on a precise mechanism.. It has been shown that the more oxidative stress is placed on the system, the more likely it is that mutations will occur. Additionally, catalase reduces mutations, whereas copper and nickel increase mutations by increasing reduction of peroxides. Some researchers believe that the oxidative stress on specific loci results in DNA polymerase pausing at those sites, creating an environment for DNA slippage to occur.[7]
Researchers first believed that MSI was random, but there is evidence suggesting that MSI targets include a growing list of genes. Examples include the transforming growth factor Beta receptor gene and the BAX gene. Each target leads to different phenotypes and pathologies.[8]
Clinical significance
Microsatellite instability may result in colon cancer, gastric cancer, endometrium cancer, ovarian cancer, hepatobiliary tract cancer, urinary tract cancer, brain cancer, and skin cancers. MSI is most prevalent as the cause of colon cancers. Each year, there are over 500,000 colon cancer cases worldwide. Based on findings from over 7,000 patients stratified for MSI-High (MSI-H), MSI-Low (MSI-L), or Microsatellite Stable (MSS) colon cancers, those with MSI-H had a more positive prognosis by 15% compared to MSI-L or MSS tumors.[9]
Colorectal tumors with MSI are found in the right colon, associated with poor differentiated tissue, high mucinogens, tumor infiltrating lymphocytes, and a presence of a Crohn's-like host response. MSI-H tumors contributing to colorectal cancer exhibit less metastasis than other derived colorectal cancer. This is demonstrated by previous research showing that MSI-H tumors are more representative in Stage II rather than Stage III cancers.[3]
Scientists have explored the connection of vacuolar protein sorting (VPS) proteins to MSI. Like MSI, VPS is linked to gastric and colon cancers. One study reports that VPS proteins were linked to MSI-H cancers, but not MSI-L cancers, thus restricting VPS to MSI-H-specific cancers.[10]
Furthermore, researchers agree that MSI-H status indicates the diagnosis of Lynch syndrome, non-metastatic prognosis, and less aggressive treatment regimens. Lynch syndrome is caused by MSI and increases the risk for colon, endometrium, ovary, stomach, small intestine, hepatobiliary tract, urinary tract, brain, and skin cancers.[3]
One study conducted over 120 Lynch syndrome patients attributing Crohn's like reaction (CLR) associated with MSI to "tumor specific neopeptides generated during MSI-H carcinogenesis." This study further corroborated that the "presence of antimetastatic immune protection in MSI-H CRC patients may explain recent findings that adjuvant 5-FU chemotherapy has no beneficial or even adverse effects in this collective." The researchers assume that there is a protective role of lymphocytes against the MSI-H CRC that prevents tumor metastasis.[11]
MSI tumors in 15% of sporadic colorectal cancer result from the hypermethylation of the MLH 1 gene promoter, whereas MSI tumors in Lynch syndrome are caused by germline mutations in MLH 1, MSH 2, MSH 6, and PMS2.[3]
MSI has been evident in the cause of sebaceous carcinomas. Sebaceous carcinomas are a subset of a larger pathology, Muir-Torre syndrome. MSI is variably expressed in Muir-Torre syndrome, most often expressed with shared pathologies in patients with colon cancer. Furthermore, MMR proteins MSH 1, MSH 2, MSH6, and PMS2 are instrumental in periocular sebaceous carcinoma, which is seen on the eyelid in 40% of sebaceous carcinomas.[12]
Microsatellite instability diagnostics
MSI is a good marker for determining Lynch syndrome and determining a prognosis for cancer treatments. The NCI has agreed on five microsatellite markers necessary to determine MSI presence: two mononucelotides, BAT25 and BAT26, and three dinucelotide repeats, D2S123, D5S346, and D17S250. MSI-H tumors result from MSI of greater than 30% of unstable MSI biomarkers. MSI-L tumors result from less than 30% of unstable MSI biomarkers. MSI-L tumors are classified as tumors of alternative etiologies. Several studies demonstrate that MSI-H patients respond best to surgery alone, rather than chemotherapy and surgery, thus preventing patients from needlessly experiencing chemotherapy.[3]
Direct and indirect mechanisms contribute to chemotherapy resistance. Direct mechanisms include pathways that metabolize the drug, while indirect mechanisms include pathways that respond to the chemotherapy treatment. The NER DNA repair pathway plays a substantial role in reversing cell damage caused by chemotherapeutic agents such as 5-FU.[13]
Discoveries since 2010
Researchers have found another MSI, called elevated microsatellite alterations at selected tetranucleotide repeats (EMAST). However, EMAST is unique in that it is not derived from MMR, and it is commonly associated with P53 mutations.[5]
EMAST is seen in a variety of cancers including those of the lung, head and neck, colorectal, skin, urinary tract, and the reproductive organs. External organ sites have more potential for EMAST. Some researchers believe that EMAST maybe a consequence of mutagenesis. EMAST positive margins in otherwise negative cancer margins suggest disease relapse for patients.[14]
References
- ↑ Ehrlich, Melanie, ed. (2000). DNA alterations in cancer: genetic and epigenetic changes. Natick, MA: Eaton Publ. p. 178. ISBN 9781881299196. Retrieved 19 February 2015.
- ↑ http://web.udl.es/usuaris/e4650869/docencia/segoncicle/genclin98/recursos_classe_(pdf)/revisionsPDF/MicrosatInstab.pdf
- 1 2 3 4 5 Buecher, B.; Cacheux, W.; Rouleau, E.; Dieumegard, B.; Mitry, E.; Lièvre, A. (2013). "Role of microsatellite instability in the management of colorectal cancers". Digestive and Liver Disease. 45 (6): 441–449. doi:10.1016/j.dld.2012.10.006.
- 1 2 Ellegren, H (2004). "Microsatellites: Simple sequences with complex evolution". Nature Reviews Genetics. 5 (6): 435–45. doi:10.1038/nrg1348. PMID 15153996.
- 1 2 Boland, C. R.; Goel, A. (2010). "Microsatellite Instability in Colorectal Cancer". Gastroenterology. 138 (6): 2073–2087.e3. doi:10.1053/j.gastro.2009.12.064. PMC 3037515
 . PMID 20420947.
. PMID 20420947. - ↑ Li, Y. C.; Korol, A. B.; Fahima, T; Nevo, E (2004). "Microsatellites within genes: Structure, function, and evolution". Molecular Biology and Evolution. 21 (6): 991–1007. doi:10.1093/molbev/msh073. PMID 14963101.
- ↑ Jackson, A. L.; Chen, R; Loeb, L. A. (1998). "Induction of microsatellite instability by oxidative DNA damage". Proceedings of the National Academy of Sciences of the United States of America. 95 (21): 12468–73. doi:10.1073/pnas.95.21.12468. PMC 22854
 . PMID 9770509.
. PMID 9770509. - ↑ Imai, K; Yamamoto, H (2008). "Carcinogenesis and microsatellite instability: The interrelationship between genetics and epigenetics". Carcinogenesis. 29 (4): 673–80. doi:10.1093/carcin/bgm228. PMID 17942460.
- ↑ Popat, S; Hubner, R; Houlston, R. S. (2005). "Systematic review of microsatellite instability and colorectal cancer prognosis". Journal of Clinical Oncology. 23 (3): 609–18. doi:10.1200/JCO.2005.01.086. PMID 15659508.
- ↑ An, C. H.; Kim, Y. R.; Kim, H. S.; Kim, S. S.; Yoo, N. J.; Lee, S. H. (2012). "Frameshift mutations of vacuolar protein sorting genes in gastric and colorectal cancers with microsatellite instability". Human Pathology. 43: 40–47. doi:10.1016/j.humpath.2010.03.015.
- ↑ Buckowitz, A; Knaebel, H. P.; Benner, A; Bläker, H; Gebert, J; Kienle, P; von Knebel Doeberitz, M; Kloor, M (2005). "Microsatellite instability in colorectal cancer is associated with local lymphocyte infiltration and low frequency of distant metastases". British Journal of Cancer. 92 (9): 1746–53. doi:10.1038/sj.bjc.6602534. PMC 2362037
 . PMID 15856045.
. PMID 15856045. - ↑ Rajan Kd, A.; Burris, C.; Iliff, N.; Grant, M.; Eshleman, J. R.; Eberhart, C. G. (2014). "DNA Mismatch Repair Defects and Microsatellite Instability Status in Periocular Sebaceous Carcinoma". American Journal of Ophthalmology. 157 (3): 640–647.e2. doi:10.1016/j.ajo.2013.12.002.
- ↑ Duldulao, M. P.; Lee, W.; Le, M.; Chen, Z.; Li, W.; Wang, J.; Gao, H.; Li, H.; Kim, J.; Garcia-Aguilar, J. (2012). "Gene Expression Variations in Microsatellite Stable and Unstable Colon Cancer Cells". Journal of Surgical Research. 174: 1–6. doi:10.1016/j.jss.2011.06.016.
- ↑ Merritt, M. A.; Cramer, D. W.; Missmer, S. A.; Vitonis, A. F.; Titus, L. J.; Terry, K. L. (2014). "Dietary fat intake and risk of epithelial ovarian cancer by tumour histology". British Journal of Cancer. 110 (5): 1392–1401. doi:10.1038/bjc.2014.16.
- Duval, A; Hamelin, R (2002). "Genetic instability in human mismatch repair deficient cancers". Annales de Génétique. 45 (2): 71–5. doi:10.1016/s0003-3995(02)01115-2. PMID 12119215.