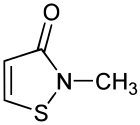
Methylisothiazolinone
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methyl-1,2-thiazol-3(2H)-one | |
| Other names
2-Methylisothiazol-3(2H)-one 2-Methyl-4-isothiazolin-3-one | |
| Identifiers | |
| 2682-20-4 | |
| 3D model (Jmol) | Interactive image |
| Abbreviations | MIT |
| ChEBI | CHEBI:53620 |
| ChemSpider | 36393 |
| ECHA InfoCard | 100.018.399 |
| EC Number | 220-239-6 |
| PubChem | 39800 |
| UNII | 229D0E1QFA |
| |
| |
| Properties | |
| C4H5NOS | |
| Molar mass | 115.1 g/mol |
| Hazards | |
| S-phrases | (S2) S26 S28 S36/37/39 S45 S60 S61 |
| R/S statement | R23/24/25 R34 R43 R50/53 |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| | |
| Infobox references | |
Methylisothiazolinone, MIT, or MI, (sometimes erroneously called methylisothiazoline), is a powerful synthetic biocide and preservative within the group of isothiazolinones, which is used in numerous personal care products and a wide range of industrial applications.
It is a cytotoxin that may affect different types of cells. Its use for a wide range of personal products for humans, such as cosmetics, lotions, moisturizers, sanitary wipes, shampoos, and sunscreens, more than doubled during the first decade of the twenty-first century and has been reported as a contact sensitizing agent by the European Commission’s Scientific Committee on Consumer Safety.
Industrial applications also are quite wide ranging, from preservative and sanitizing uses to antimicrobial agents, energy production, metalworking fluids, mining, paint manufacturing, and paper manufacturing, many of which increase potential exposure to it by humans as well as organisms, both terrestrial and marine. Industrial applications in marine environments are proving to be toxic to marine life, for instance, when the effect of its now almost-universal use in boat hull paint was examined.[1]
Applications
Methylisothiazolinone and other isothiazolinone-derived biocides are used for controlling microbial growth in water-containing solutions.[2] Two of the most widely used isothiazolinone biocides are 5-chloro-2-methyl-4-isothiazolin-3-one (chloromethylisothiazolinone or CMIT) and 2-methyl-4-isothiazolin-3-one (methylisothiazolinone or MIT), which are the active ingredients in a 3:1 mixture (CMIT:MIT) sold commercially as Kathon. Kathon is supplied to manufacturers as a concentrated stock solution containing from 10-15% of CMIT/MIT. For applications the recommended use level is from 6 ppm to 75 ppm active isothiazolones. Biocidal applications range from industrial water storage tanks to cooling units, in processes as varied as mining, paper manufacturing, metalworking fluids and energy production.
Kathon also has been used to control slime in the manufacture of paper products that contact food. In addition, this product serves as an antimicrobial agent in latex adhesives and in paper coatings that also contact food.[3]
Other isothiazolinones
One isothiazolinone, Sea-Nine 211 (4,5-dichloro-2-n-octyl-4-isothiazolino-3-one, DCOI), has quickly replaced tributyltin as the antifouling agent of choice in ship hull paint. A recent study reported the presence of DCOI in both port water and sediment samples in Osaka, Japan, especially in weakly circulating mooring areas.[4] Of environmental concern, DCOI levels predicted in marinas now are considered a threat to various marine invertebrate species.[5] Isothiazolinones also are extremely toxic to fish.[1]
In industrial use, the greatest occupational inhalation exposure occurs during open pouring. Non-occupational exposure to isothiazolinones by the general population also occurs, albeit at much lower concentrations.[6] These compounds are present in a very large number of commonly used cosmetics.[7] “Leave-on” cosmetics (hand-creams, lotions, etc.) contain 15 parts per million (100 micromolar) of combined CMIT/MIT.
Human health
MIT is allergenic and cytotoxic, and this has led to some concern over its use.[8][9] A report released by the European Scientific Committee on Cosmetic Products and Non-food Products Intended for Consumers (SCCNFP) in 2003 also concluded that insufficient information was available to allow for an adequate risk assessment analysis of MIT.[10]
Rising reports of consumer impact led to new research, including a report released in 2014 by the European Commission Scientific Committee on Consumer Safety which reported: "The dramatic rise in the rates of reported cases of contact allergy to MI, as detected by diagnostic patch tests, is unprecedented in Europe; there have been repeated warnings about the rise (Gonçalo M, Goossens A. 2013). The increase is primarily caused by increasing consumer exposure to MI from cosmetic products; exposures to MI in household products, paints and in the occupational setting also need to be considered. The delay in re-evaluation of the safety of MI in cosmetic products is of concern to the SCCS; it has adversely affected consumer safety."[11]
"It is unknown what proportion of the general population is now sensitized to MI and has not been confirmed as sensitized."[11] In 2014, the European Commission Scientific Committee on Consumer Safety further issued a voluntary ban on "the mixture of Methylchloroisothiazolinone (and) Methylisothiazolinone (MCI/MI) from leave-on products such as body creams. The measure is aimed at reducing the risk from and the incidence of skin allergies. The preservative can still be used in rinse-off products such as shampoos and shower gels at a maximum concentration of 0.0015 % of a mixture in the ratio 3:1 of MCI/MI. The measure will apply for products placed on the market after 16 July 2015."[12] Shortly thereafter, Canada moved to adopt similar measures in its Cosmetic Ingredients Hotlist.[13] Additionally, new research into cross reactivity of MI-sensitized patients to variants benzisothiazolinone and octylisothiazolinone have found that reactions may occur if present in sufficient amounts.[14]
Allergic contact dermatitis
Methylisothiazolinone is used commonly in products in conjunction with methylchloroisothiazolinone, a mixture sold under the registered trade name Kathon CG. A common indication of sensitivity to Kathon CG is allergic contact dermatitis. Sensitization to this family of preservatives was observed as early as the late 1980s.[15][16] Due to increased use of isothiazolinone-based preservatives in recent years, an increase in reported incidences of contact allergy to this product has been reported. In 2013 the substance was declared the 2013 Contact Allergen of the Year by the American Contact Dermatitis Society.[17] In 2016 the Dermatitis Academy launched a call to action for patients to report their isothiazolinone allergy to the FDA.[18]
On December 13, 2013 the trade group, Cosmetics Europe,[19] following discussions with the European Society of Contact Dermatitis (ESCD),[20] recommended to its members "that the use of Methylisothiazolinone (MIT) in leave-on skin products including cosmetic wet wipes is discontinued. This action is recommended in the interests of consumer safety in relation to adverse skin reactions. It is recommended that companies do not wait for regulatory intervention under the Cosmetics Regulation but implement this recommendation as soon as feasible."[21]
On March 27, 2014, the European Commission’s Scientific Committee on Consumer Safety issued an opinion on the safety of Methylisothiazolinone. This report only considered the issue of contact sensitization. The committee concluded:
“Current clinical data indicate that 100 ppm MI in cosmetic products is not safe for the consumer.
"For leave-on cosmetic products (including ‘wet wipes’), no safe concentrations of MI for induction of contact allergy or elicitation have been adequately demonstrated.
"For rinse-off cosmetic products, a concentration of 15 ppm (0.0015%) MI is considered safe for the consumer from the view of induction of contact allergy. However, no information is available on elicitation.”[22]
Ingredient in mouthwash
Although many companies that had begun using the biocide in personal care products, such as Kimberly-Clark, Johnson and Johnson, and Unilever are removing methylisothiazolinone from their lotions and wipes, Colgate-Palmolive has added it as an ingredient in a mouthwash put onto the market in August 2014 with the name Colgate Total Lasting White.[23] The New York Times reported that some companies even are considering removing the biocide from products that routinely are rinsed off shortly after application.
References
- 1 2 Rohm and Haas, Toxicology Department, “Evaluation of the toxicity of Kathon biocide,” August, 1984
- ↑ Collier PJ. Ramsey A. Waigh RD. Douglas KT. Austin P. Gilbert P.; Ramsey; Waigh; Douglas; Austin; Gilbert (1990). "Chemical reactivity of some isothiazolone biocides". Journal of Applied Bacteriology. 69 (4): 578–584. doi:10.1111/j.1365-2672.1990.tb01551.x. PMID 2292521.
- ↑ "Cosmetic Ingredient Review. Final Report on the Safety Assessment of Methylisothiazolinone and Methylchloroisothiazolinone". J. American Col. Toxicol. 11 (1): 75–128. 1992. doi:10.3109/10915819209141993.
- ↑ Harino H. Mori Y. Yamaguchi Y. Shibata K. Senda T.; Mori; Yamaguchi; Shibata; Senda (2005). "Monitoring of antifouling booster biocides in water and sediment from the port of Osaka, Japan". Arch Environ Contam Toxicol. 48 (3): 303–310. doi:10.1007/s00244-004-0084-2. PMID 15750770.
- ↑ Bellas J. (2006). "Comparative toxicity of alternative antifouling biocides on embryos and larvae of marine invertebrates". Sci Total Environ. 367 (2–3): 573–85. doi:10.1016/j.scitotenv.2006.01.028. PMID 16545431.
- ↑ Environmental Protection Agency (1998). R.E.D. Facts, Methylisothiazolinone. Publication EPA-738-F-98-008
- ↑ Rastogi SC, SC (1990). "Kathon CG and cosmetic products". Contact Dermatitis. 22 (3): 155–160. doi:10.1111/j.1600-0536.1990.tb01551.x. PMID 2335088.
- ↑ A. Schnuch, J. Geier, W. Utur, P. J. Frosch: "Patch testing with preservatives, antimicrobials and industrial biocides. Results from a multicentre study", British Journal of Dermatology, 137(3), 467-476 (1998).
- ↑ A. C. De Groot, A. Herxheimer: "Isothiazolinone Preservative: Cause Of A Continuing Epidemic Of Cosmetic Dermatitis", The Lancet, Volume 333, Issue 8633, Pages 314-316 (1989).
- ↑ European Scientific Committee on Cosmetic Products and Non-food Products Intended for Consumers (SCCNFP), adopted 2004
- 1 2 SCCS (Scientific Committee on Consumer Safety), Opinion on Methylisothiazolinone (P94) – Submission II, 12 December 2013, SCCS/1521/13, revision of 27 March 2014 (PDF)
- ↑ "European Commission - PRESS RELEASES - Press release - Consumers: Commission improves safety of cosmetics". europa.eu. Retrieved 2016-08-10.
- ↑ "Health Canada - "December 2015 Changes to the Cosmetic Ingredient Hotlist"".
- ↑ Schwensen, J. F.; Menné Bonefeld, C.; Zachariae, C.; Agerbeck, C.; H.Petersen, T; Geisler, C.; E.Bollmann, U; Bester, K.; D.Johansen, J (2016-06-01). "Cross-reactivity between methylisothiazolinone, octylisothiazolinone and benzisothiazolinone using a modified local lymph node assay". British Journal of Dermatology: n/a–n/a. doi:10.1111/bjd.14825. ISSN 1365-2133.
- ↑ Hannuksela, M. (1986). "Rapid increase in contact allergy to Kathon CG in Finland". Contact dermatitis. 15 (4): 211–214. doi:10.1111/j.1600-0536.1986.tb01338.x. PMID 3802805.
- ↑ De Groot, A. C.; Weyland, J. W. (1988). "Kathon CG: A review". Journal of the American Academy of Dermatology. 18 (2 Pt 1): 350–358. doi:10.1016/s0190-9622(88)70051-1. PMID 3279090.
- ↑ Castanedo-Tardana, M. P.; Zug, K. A. (2013). "Methylisothiazolinone". Dermatitis : contact, atopic, occupational, drug. 24 (1): 2–6. doi:10.1097/DER.0b013e31827edc73. PMID 23340392.
- ↑ MCI/MI/BIT Call To Action!
- ↑ Cosmetics Europe Recommendation on MIT
- ↑ European Society of Contact Dermatitis
- ↑ Cosmetics Europe Recommendation on MIT
- ↑ European Commission Scientific Committee on Consumer Safety Opinion on Methylisothiazolinone (P94) Submission II (Sensitization only) SCCS/1521/13
- ↑ Abrams, Rachel, Growing Scrutiny for an Allergy Trigger Used in Personal Care Products, The New York Times, January 23, 2015, and in the print edition of the New York edition, as An Unexpected Reaction, January 24, 2015, p. B1
External links
- 2014 EPA Re-registration Review Docket with Public Commentary
- Reregistration Eligibility Decision of MIT by US EPA
- Record in the Household Products Database of NLM
- Material Safety Data Sheet for product containing 0.1-1% MIT
- Commission Scientific Committee on Consumer Safety Opinion on Methylisothiazolinone (P94) Submission II (Sensitization only) SCCS/1521/13
- Methylisothiazolinone at the National Library of Medicine
- 2014 EPA Re-registration Review Docket with Public Commentary
- Photo album of self reported reactions to methylisothiazolinone