Lymantria dispar dispar
| Gypsy moth | |
|---|---|
| | |
| Adult female gypsy moth | |
 | |
| Adult male gypsy moth | |
| Scientific classification | |
| Kingdom: | Animalia |
| Phylum: | Arthropoda |
| Class: | Insecta |
| Order: | Lepidoptera |
| Family: | Erebidae |
| Subfamily: | Lymantriinae |
| Genus: | Lymantria |
| Species: | L. dispar |
| Subspecies: | L. d. dispar |
| Trinomial name | |
| Lymantria dispar dispar (Linnaeus, 1758) | |
Lymantria dispar dispar, commonly known as the gypsy moth, European gypsy moth, or North American gypsy moth, is a moth in the family Erebidae that is of Eurasian origin. It has a range which covers Europe, Africa, and North America.
Carl Linnaeus first described the species Lymantria dispar in 1758. The subject of classification has changed throughout the years, resulting in confusion surrounding the species' taxonomy. This caused many references to describe this one species in different ways. The family has jumped between Lymantriidae, Noctuidae and Erebidae. Lymantria dispar dispar has been designated a sub-species of Lymantria dispar.
It is classified as a pest, and its larvae consume the leaves of over 500 species of trees, shrubs and plants. The gypsy moth is one of the most destructive pests of hardwood trees in the eastern United States.
Taxonomy
Linnaeus first described the species Lymantria dispar in 1758.[1]:2 The subject of classification has changed throughout the years, resulting in confusion surrounding the species taxonomy. This caused many references to describe this one species in different ways. The family has jumped between Lymantriidae, Noctuidae and Erebidae. The species Lymantria dispar has been split into sub-species such as Lymantria dispar asiatica and Lymantria dispar japonica. Differences between the subspecies are poorly understood even at the government level.
Order
The order Lepidoptera contains moths and butterflies characterized by having a complete metamorphosis; larvae transform to pupae and then metamorphose into adult moths or butterflies.[2]:9 The family is Lymantriidae.[2]:9 Lymantriid larvae are commonly called tussock moths because of the tufts of hair on the larvae.[2]:9
Classified as Lymantriidae
Lymantriidae was separated from Lariidae and was classified as its own family by (Hampson 1893).[3] The family Lymantriidae had previously been referred to as Orgyiidae (Wallengren 1861) and Dasychiridae (Packard 1864)), but consensus formed in favor of Lymantriidae.[3] Lymantriidae has had numerous taxonomical changes which was solidified by Ferguson in 1978.[3]
Reclassified as Noctuidae
Lymantriidae was reclassified into the family Noctuidae in 2006.[1]:2 Noctuidae is defined as a group in which hindwing vein M2, originates in the lower third of the discal cell, but it is as strong as vein M3.[1]:2 This results in the cubital vein appearing to have four branches.[1]:2 The classification of Noctuidae came from Mitchel et al. (2005).[1]:2 Lafontaine and Fibiger (2006) agreed that Lymantriidae belonged within Noctuidae, but disagree with the classification as a subfamily.[1]:2 Fewer taxonomic changes are required with the reclassification of Lymantriidae being a subfamily in the monophyletic group of Noctuidae.[1]:2 Sources published prior to the reclassification in 2006 and shortly thereafter may refer to Lymantriidae.[4] Lafontaine and Fibiger (2006)'s classification as a subfamily of Noctuidae has received acceptance, but is not yet universal.[1]:2[5][6][7]
Reclassified as Erebidae
In 2012, Lymantriidae was moved from Noctuidae to Erebidae, further complicating the taxonomy.[8] The newly created family has been reflected in some sources.[9]
Etymology
The name Lymantria dispar is composed of two Latin-derived words. Lymantria means 'destroyer'.[10] The word dispar is derived from the Latin for 'unequal' and it depicts the differing characteristics between the sexes.[2]:9
L. d. dispar versus L. dispar usage
For the purposes of taxonomy, the common names North American gypsy moth and European gypsy moth represent the same subspecies, Lymantria dispar dispar, as opposed to subspecies of Lymantria dispar from Asia.[1]:6
Confusion over the species and subspecies is widespread. The U. S. Department of Agriculture defines the Asian gypsy moth as "any biotype of Lymantria dispar possessing female flight capability",[1]:5 despite Lymantria dispar asiatica not being the only classified subspecies that is capable of flight.[1]:6 Traditionally, Lymantria dispar have been referred to as "gypsy moths" even when referring to Japanese, Indian, and Asiatic gypsy moths.[1]:5
Range
The gypsy moth, Lymantria dispar dispar, is indigenous to Europe, and was introduced in North America.[1]:5 Though the Asiatic and the Eurasian moths were not classified as different subspecies in the 1980s, the differences were already notable.[2]:9 The species was considered extinct in the UK from soon after 1900, but colonies have been found in London from 1995 and Buckinghamshire from 2005.[11][12]
Introduction to North America
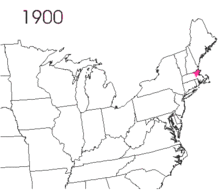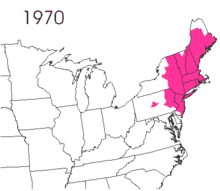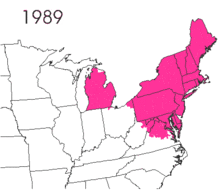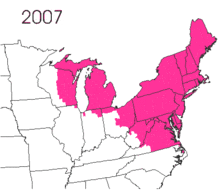
The gypsy moth was introduced into North America in 1869 from Europe.[2]:9 Étienne Léopold Trouvelot imported the moths, with the intent of interbreeding gypsy moths with silk worms to develop a silkworm industry.[2]:10 The moths were accidentally released from his residence in Medford, Massachusetts.[2]:10 There are conflicting reports on the resulting actions. One states that despite issuing oral and written warnings of possible consequences, no officials were willing to assist in searching out and destroying the moths.[2]:10 The other notes that he was aware of the risk and there is no direct evidence that he contacted government officials.[1]:1
As noted in The Gypsy Moth (1896) by Forbush and Fernald, the gypsy moth was considered a nuisance just ten years after their release.[2]:10 The first major outbreak occurred in 1889, and Forbush and Fernald recount the extent of devastation: all the trees being defoliated and caterpillars covering houses and sidewalks and raining down upon residents.[2]:1,10 At first it was uncertain what species was responsible for the outbreak, but after the caterpillar was identified by entomologist Maria Elizabeth Fernald, an eradication program began in 1890.[1]:1[13]
Spread
The small larvae of the gypsy moth take to the air and are carried by the wind.[2]:10 The larvae spin silken threads and hang from them, waiting for the wind to blow.[2]:10 The light larvae have long hairs that increase their surface area, which are suitable for being carried aloft.[2]:10 The natural spread is slow, but transportation of the moth has led to isolated gypsy moth populations, with accidental transport of the eggs being noted.[2]:10 According to the United States Department of Agriculture, without intervention, this pest spreads about 13 miles per year.[14] Typically, short distances can be traversed by larva, but there is suspicion that long distance flights are possible. It has been hypothesized that storms carried the larva across Lake Michigan to the western shore, a span of dozens of miles.[15]
Firewood is a common way to transport eggs, since the moths will lay their eggs on dead wood.[16] Firewood should be purchased and used locally, within 50 miles of where it is cut, to prevent the spread of the moths.[16] The website Don't move Firewood offers useful information for Americans about the pests, like the gypsy moth, that can be transported specifically by firewood.[17]
Life cycle
Eggs
Gypsy moth egg masses are typically laid on branches and trunks of trees, but may be found in any sheltered location, including rocks, foliage and vehicles.[2]:12 Female gypsy moths are flightless, so they lay eggs on a surface near where they emerged from their pupa.[2]:12 The eggs are covered in a coating of hairs.[2]:12 The covering provides protection from predators and parasites and may be important for insulating the eggs from cold temperatures and sealing in moisture.[2]:12
The larva inside the egg becomes fully developed in about one month.[2]:13 The egg is the overwintering stage lasting for eight or nine months.[2]:13 Development ceases in preparation for the winter.[2]:13 After an acclimation stage, during which the larva inside the egg reduces its water content, eggs can withstand freezing temperatures.[2]:13 The larva inside the egg resumes activity in the spring, and reabsorbs water.[2]:13 The larva will then chew through the chorion of the egg and the protective hair of the egg cluster in the spring.[2]:13
Egg clusters are usually an oval about 3/4 of an inch wide and 1.5 inches long.[18] The egg masses are a buff yellow-brown color, likened to a manila folder, but may bleach out over the winter months.[19] As the female lays them, she covers them with hair-like setae from her abdomen. Egg clusters contain from 100 to 1000 eggs.[2]:12 Due to the appearance of the eggs, several common names have emerged; Schwammspinner is German for "fungus spinner" and la spongieuse is French for the texture of the egg cluster.[2]:12
Larvae

Larvae (caterpillars) emerge from egg masses in the spring.[2]:13 Most larvae hatch within a week, but it could take as long as a month.[2]:13 The new larvae remain on or near the egg cluster if they hatch in rainy weather or if the temperature is below 7C.[2]:13 The larva will disperse even if there is enough foliage for growth, they hang from silk threads and wait for the wind to send them aloft.[2]:13 The larva are about 3 mm when they first hatch and will grow to 50 or 90 mm in size.[2]:14
The larva will first feed on the leaf hairs and then move onto the leaf epidermis.[2]:14 Feeding occurs in the daytime, primarily in the morning and late afternoon.[2]:14 As the larva grow, the feeding becomes a nocturnal activity.[2]:14 When not eating, the larva will remain on the underside of the leaf and make a mat of silk for attachment.[2]:14

To grow, the larva must molt.[2]:14 Larva are characterized by the term 'instar', which refers to the number of times a larva has molted; a first-instar has not yet molted, a second-instar has molted once, a third-instar has molted twice, etc.[2]:15 Males typically are five instars and females are six instars.[2]:15 When the larvae reach the fourth instar, they become nocturnal feeders, and will return to their resting places at dawn, hiding under flaps of bark, in crevices, or under branches - any place that provides protection.[2]:15
Newly hatched larvae are black with long, hair-like setae. Older larvae have five pairs of raised blue spots and six pairs of raised brick-red spots along their backs, and a sprinkling of setae.[2]:15 As the larval stage comes to an end they cease feeding and surround themselves in a silken net.[2]:15
Distinguishing the larvae from other species
Larvae can be distinguished from other species of caterpillar by its spots. Close to the head, 5 pairs of blue spots and 6 pairs of red spots to its tail.[20] This distinction will prevent confusion with the Spiny elm caterpillar and other caterpillars.[20] Tent Caterpillars are one example, which can be distinguished by its silky tents in trees, a stripe on its back, and its preference for cherry trees.[21]
Pupae
.jpg)
The larvae reach maturity between mid-June and early July, then enter the pupal stage, during which larvae change into adult moths. Pupation lasts from seven to 14–17 days.[2]:16 The adult moth will emerge, fully developed, by splitting the pupal skin.[2]:16
When the population is spread out and running low, pupation can take place under flaps of bark, in crevices, under branches, on the ground, and in other places where larvae rested. During periods when population numbers are dense, pupation is not restricted to these locations, but can take place in sheltered and open locations, even exposed on the trunks of trees or on foliage of nonhost trees. Usually, the caterpillars create flimsy cocoons made of silk strands holding the leaf together, while others do not cover their pupae in cocoons, but rather hang from a twig or tree bark, like butterfly pupae do.
Adults

The males have feathery antennae versus the thin antennae of females.[2]:11 Size differences are also noted, with the forewing of the male moth being 20–24 mm long, and that of the female 31–35 mm.[22] Obviously, from this it is noticeable that the females are bigger than the males. Another important difference between the sexes is that females possess fully formed wings, but do not fly.[2]:11–12 Female flight is common in Eurasia and Asia, but these moths may be of a different species.[2]:11–12 The other species is called the Asian Gypsy Moth.
Moths usually emerge from pupae in July, but it can vary with population density and climate.[2]:11 The brown male gypsy moth emerges first, usually one or two days before the females do.[2]:11 The males fly in rapid zigzag patterns, but are capable of direct flight.[2]:11 Like most moths, the males are typically nocturnal, but can sometimes be seen flying during the day as well. The males fly up and down tree trunks, or other vertical objects in search of females.[2]:11 When heavy, black-and-white egg-laden females emerge, emitting a pheromone that attracts the males.[2]:12 The female has a small gland near the tip of the abdomen which releases the pheromone, with a pumping motion, termed 'calling'.[2]:12 It can attract males from long distances, tracking the scent through its erratic flight pattern.[2]:12
Courtship is not elaborate; the female must raise her wing to allow the male to couple with her.[2]:12 The moths remain in copula for up to an hour, but the transfer of the spermatophore is usually accomplished within 10 minutes.[2]:12 Male moths can inseminate more than one female.[2]:12 Multiple mating in females is possible, but uncommon, because the female stops releasing the attracting pheromone after mating.[2]:12 After mating, the females begin depositing the eggs.[2]:12
The adult moths live about one week.[2]:12 They do not possess an active digestive system and cannot feed, but can drink in moisture.[2]:12 The reproductive chance for females lasts about two days, with the pheromone for attracting males being diminished by the third day.[2]:12 Due to the pheromone's potency, most females will mate.[2]:12
The females also produce offspring after they mate with a male, laying eggs on trees, shrubs, rocks, vehicles, and plants of many types. She typically lays about 500 eggs. The eggs are covered with a peachy fuzz that can cause serious rashes if they are touched by bare skin or fur, especially on humans and mammals. Then the female leaves to eat, while her eggs are protected. She does not live to see her offspring.[2]:12
Behavior
Many environmental factors such as resource availability, predator density and sexual competition are known to affect gypsy moth behavior from its larval stage to adult stage. Population of gypsy moth will exist for many years in low densities. When the population enters the release phase, the population rapidly expands to the outbreak phase where the population size will rise several magnitude and fall back within only a few generations.[23]
Feeding
The gypsy moth brings one of the largest impacts in defoliation of deciduous trees in the Northern Hemisphere. Since its introduction in to the United States in 1868 or 1869, it has spread both West and South, now taking over most of the hardwood forests in the eastern United States and Canada [23] Over three hundred species of trees and shrubs are host to the gypsy moth.[2]:16 Gypsy moth larvae prefer oak trees, but may feed on many species of trees and shrubs, both hardwood and conifer.[2]:16
In the eastern US, the gypsy moth prefers oaks, aspen, apple, sweetgum, speckled alder, basswood, gray, paper birch, poplar, willow, and hawthorns, among other species.[24] Older larvae feed on several species of softwood that younger larvae avoid, including cottonwood, hemlock, Atlantic white cypress, and pine and spruce species native to the east.[24] The gypsy moth avoids ash trees, tulip-tree, American sycamore, butternut, black walnut, catalpa, flowering dogwood, balsam fir, cedar, American holly, and mountain laurel and rhododendron shrubs, but will feed on these in late instars when densities are extremely high.[24]
Larvae will climb up any object in their path in search of food, including telephone poles, vehicles, even people.[2]:16
Predation
Many species have been identified as preying on L. dispar. Some species, such as the white-footed mouse or Anastaus disparis, have a significant impact on the gypsy moth population dynamics. On the other hand, avian predation and invertebrate predation show only small effects on the population dynamics of the gypsy moth.
Small mammals
The white-footed mouse, Peromyscus leucopus, is considered important for regulating sparse gypsy moth populations.[2]:21 Rodents consume larvae and pupae that seek resting sites near or on the ground.[2]:21 The white-footed mouse is the most common and widely distributed small mammal in the US Northeast.[2]:108 The shorttail shrew is common east of the Rocky Mountains and will consume the larva and pupa.[2]:108
Birds
Insectivorous birds prey upon the larva of the gypsy moth, but the egg clusters are protected by their hair coverings.[2]:21 The effects of bird predation have not been fully studied in North America,[2]:21 but it has been well-documented in Japan and Eurasia.[2]:21 When outbreaks of gypsy moths occur, bird predation has no significant effect on the population.[2]:105 Birds that consume gypsy moth larvae, pupae and adults include the bluejay, red-eyed vireo, rufous-sided towhee, northern oriole, catbird and the robin.[2]:105 The black-capped chickadee preys upon the entire life-cycle, including the eggs.[2]:105
Many bird species feed on gypsy moth larvae, but they are not a major food in the diet of any common bird species. Although a few European studies cite avian predation as a large influence in keeping the gypsy moth population in control, few studies exist to prove it.[23]
Invertebrate predation
Calosoma sycophanta is a beetle that preys upon the gypsy moth larvae and pupae. Larvae and adults of the species tear open their prey and feed upon them.[2]:21 In low density populations, there is a positive correlation between larval mortality and predation rates on pupae.[23]
Parasites
Gypsy moth parasitoids have been widely studied, but they do not seem to have major effects on the population.
Four species of parasitic flies prey on gypsy moth larva.[2]:20 Parasetigana silvestris and Exorista larvarum lays an egg on the gypsy moth larva. If that egg hatches before the gyspy moth larva molts, the fly larva will penetrate the host.[2]:20 Compsilura concinnata pierces the gypsy moth larva and deposits its own larva inside.[2]:20 Blepharipa pratensis, lays its eggs on leaves, the gypsy moth larva will consume the egg and the fly larva will hatch inside its gut.[2]:20
Eight species of parasitic wasps attack the gypsy moth. Ooencyrtus kuvanae and Anastatus disparis attack the eggs.[2]:20 Ooencyrtus kuvanae attacks the eggs, but the effectiveness is limited by the ovipositor which can only penetrate the surface layer of the egg cluster.[2]:20 Anastatus disparis has limited success as a predator because it can only attack unembroyonated eggs and the female wasps do not have wings.[2]:20Even so, Anastatus disparis is the only species that is known to occasionally impact the population dynamics of the gypsy moth. Apanteles melanoscelus and Phobocampe disparis parasitize the early larva stages.[2]:20 Brachymeria intermedia and Monodontomerus aureus parasitize the gypsy moth pupae.[2]:20
A parasite native to North America, Itoplectes conquisitor, attacks and kills gypsy moth pupae, but development of the larva is rare within the host and the number of attacks themselves is also low.[2]:20
Glyptapanteles portheriae and G. liparidis are both wasps that lay eggs on the gypsy moth larva. Study of the G. liparidis showed almost 90% success when the host is parasitized during premolt to the 3rd instar. Most invertebrate predation occurred when larvae were in the litter.[25]
The population in Austria suffered high parasitism by the tachinids Parasetigena silvestris, which may have contributed in prevention of further increase in L. dispar in this locality.[26]
Pathogens
Gypsy moth populations in different locations show vulnerability to different viral species. Nuclear polyhedrosis virus caused significant mortality in some cases. The most important pathogen is the Lymantria dispar multicapsid nuclear polyhedrosis virus (LdMNPV), sometimes referred to as NPV or Borralinivirus reprimens.[2]:21 The viral particles are consumed by the larva when eating through the egg chorion and will die in the first instar.[2]:21 The bodies disintegrate, spreading the virus on the foliage which will then be consumed by other larvae.[2]:21 The virus' mortality is dramatic in outbreaks, which causes high mortality,[2]:21 and the odor of the decaying larvae permeates the area.[2]:19 The pathogen is used as an insecticide under the name Gypchek [2]:21
Steptococcus faecalis is another pathogen which is notable for its mortality.[2]:21 Larva killed by the pathogen have a shrivelled appearance.[2]:21
Fungi
Entomophaga maimaiga a Japanese fungi also helps control the population.[27]
Other adaptations
Population density of larva plays a major role in their behavior. At low density, larva remain inactive during the day, but in high populations become hyperactive.[2]:16
Interbreeding produces deleterious genetic effects, and dispersal serves to reduces this effect.[2]:23
Weather
Temperature is important to the gypsy moth. Low temperatures are fatal. Temperatures of −9 C can be withstood during the winter, an extended period will kill the larvae inside and −23 C even for a short time is lethal.[2]:22 Egg deposits that are low or on the ground can be insulated against temperatures, including snow, and survive the lethal temperatures.[2]:22 Temperatures above 32 C increase growth and development.[2]:22 Rainfall can drown larva before they are established, low populations are correlated to heavy rainfall during the larval stage.[2]:22 Wind is also critical to the dispersal of the larva, wind speeds of several miles per hour are enough to break the silk threads and disperse the larva.[2]:23
Pheromones
Disparlure has been synthesized artificially, and has been used to confuse mating patterns or lead male moths into traps, however this technique is less successful against concentrated populations, hence its main use may be to delay infestation until other ecological solutions are found.[28]
Impact
The gypsy moth habitat overlaps with the northern tiger swallowtail, Papilio canadensis. Experiments indicate that known gypsy moth pathogens and gypsy moth bodily fluid negatively affect the survival of swallowtail larvae. Gypsy moth bodily fluid is lethal and swallowtail caterpillars were prone to higher rates of parasitism when placed in the field near gypsy moth infestations.[29]

Lymantria dispar dispar causes widespread defoliation and costs the economy millions of dollars in damages. Total defoliation in America, from 1970 to 2010, was 80.4 million acres (325,000 km2).[30] The worst year was 1981 with 12.9 million acres (52,000 km2) defoliated.[24][31] In 2010, 1,207,478 acres (488,649 ha) were defoliated.[20]
Forest defoliation by the gypsy moths each year impacts the populations and reproductive success of forest-dwelling birds. Nests placed in defoliated sites suffered a higher predation rate than those in non-defoliated sites. Gypsy moths have a direct impact on avian behavior in the American forests.[32]
References
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Pogue, Michael. "A review of selected species of Lymantria Huber [1819]" (PDF). Forest Health Technology Enterprise Team. Retrieved September 14, 2012.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 100 101 102 103 The Gypsy Moth: Research Toward Integrated Pest Management, United States Department of Agriculture, 1981
- 1 2 3 Watson, A., Fletcher, D.S. & Nye, I.W.B., 1980, in Nye, I.W.B. [Ed.], The Generic Names of Moths of the World, volume 2 © Trustees of the British Museum (Natural History), Publication Number 811 ISBN 0-565-00811-0 http://www.nhm.ac.uk/research-curation/research/projects/butmoth/pdf/GNMW_Vol_2.pdf
- ↑ "Revised higher classification of the Noctuoidea (Lepidoptera)". The Canadian Entomologist. 138: 610–635. doi:10.4039/n06-012.
- ↑ Lymantria dispar (Linnaeus, 1758) Taxonomic Serial No.: 709249
- ↑ Bugguide.net's Taxonomic data.
- ↑ NCBI Taxonomy browser
- ↑ Zhari; et al. (January 2012). "Molecular phylogenetics of Erebidae (Lepidoptera, Noctuoidea)". Systematic Entomology. Systematic Entomology. 37: 102–124. doi:10.1111/j.1365-3113.2011.00607.x. Retrieved September 15, 2012.
- ↑ Butterfliesandmoths.org Lymantria dispar classified as Erebidae
- ↑ Free Dictionary for Lymantria
- ↑ Waring, P.; et al. (2009), Field Guide to the Moths of Great Britain and Ireland, Hook, Hampshire: British Wildlife Publishing, ISBN 0953139999
- ↑ "Gypsy moth, Lymantria dispar", UK Moths, Ian Kimber, retrieved January 23, 2013
- ↑ Spear, Robert J. The Great Gypsy Moth War: The history of the first campaign in Massachusetts to eradicate the gypsy moth, 1890-1901. University of Massachusetts Press, 2005.
- ↑ "Gypsy Moth". United States Department of Agriculture. Retrieved September 14, 2012.
- ↑ KL Frank. "Interpretation of gypsy moth frontal advance using meteorology in a conditional algorithm.". Retrieved September 14, 2012.
- 1 2 Witten, Scott. "Chop local". Laurinburg Exchange. Retrieved September 14, 2012.
- ↑ http://www.dontmovefirewood.org/
- ↑ "Gypsy moth: Biology & Life Cycle". Retrieved September 14, 2012.
- ↑ "Identifying and managing Gypsy Moth egg masses" (PDF). Chris Williamson and Andrea Diss. Retrieved September 14, 2012.
- 1 2 3 "European Gypsy Moth (Lymantria dispar)" (PDF). Retrieved September 21, 2012.
- ↑ "Gypsy Moth in New Jersey" (PDF). Forest Resource Education Center New Jersey Forest Service Department of Environmental Protection. Retrieved September 21, 2012.
- ↑ (Waring et al., 2003)
- 1 2 3 4 Elkinton, J. S.; A. M. Liebhold (1990). "Population dynamics of gypsy moth in North America". Annual Review of Entomology. 35: 571–596. doi:10.1146/annurev.en.35.010190.003035.
- 1 2 3 4 M.McManus, N. Schneeberger, R. Reardon, and G. Mason (October 1989). "Forest Insect & Disease Leaflet 162 – Gypsy Moth". U.S. Department of Agriculture Forest Service. Retrieved 2010-07-10.
- ↑ Marktl, Robert C.; Christian Stauffer; Axel Schopf (2002). "Interspecific competition between the braconid endoparasitoids Glyptapanteles porthetriae and Glyptapanteles liparidis in Lymantria dispar larvae". Entomologia Experimentalis et Applicata. 105: 97–109. doi:10.1046/j.1570-7458.2002.01038.x.
- ↑ Hoch, G.; M. Zubrik; J. Novotny; A. Schopf (2001). "The natural enemy complex of the gypsy moth, Lymantria dispar in different phases of its population dynamics in eastern Austria and Slovakia - a comparative study". Journal of Applied Entomology (125): 217–227.
- ↑ Ferrechio, Susan. "Nature Saves Trees from Gypsy Moths: Fungus Helps Stop Spring Caterpillars Cold". The Washington Times (Washington, DC). Retrieved September 19, 2012.
- ↑ Gerardi, Michael H.; Grimm, James K. (1978). The history, biology, damage, and control of the gypsy moth, Porthetria dispar (L.). Rutherford [N.J.]: Fairleigh Dickinson University Press. ISBN 083862023X.
- ↑ Redman, Ahnya M.; Scriber, J. Mark (June 2000). "Competition between the gypsy moth, Lymantria dispar, and the northern tiger swallowtail, Papilio canadensis: interactions mediated by host plant chemistry, pathogens, and parasitoids". Oecologia. 125 (2): 218–228. doi:10.1007/s004420000444.
- ↑ Defoliation chart from US Forest Service
- ↑ Gypsy Moths Return to Northeast Worst Outbreak in a Decade Descends on Northeast; Entomologists Do Not Know How to Stop It. SUMMER'S MUNCH, Laurent Belsie, The Christian Science Monitor, July 2, 1990
- ↑ Thurber, Dale K.; McClain, Willam Robert; Whitmore, Robert C. (July 1994). "Indirect effects of gypsy moth defoliation on nest predation". The Journal of Wildlife Management. 58: 493–500. doi:10.2307/3809321.