Hippocampus
| Hippocampus | |
|---|---|
|
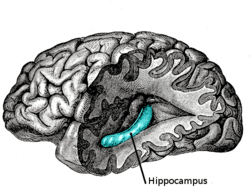
The hippocampus is located in the medial temporal lobe of the brain. In this lateral view of the human brain, the frontal lobe is at left, the occipital lobe at right, and the temporal and parietal lobes have largely been removed to reveal the hippocampus underneath. | |
|
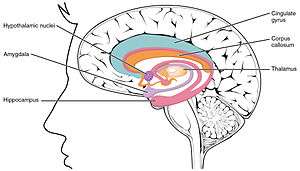
Hippocampus (lowest pink bulb) as part of the limbic system | |
| Details | |
| Part of | Temporal lobe |
| Identifiers | |
| Latin | Hippocampus |
| MeSH | Hippocampus |
| NeuroNames | hier-164 |
| NeuroLex ID | Hippocampus |
| TA | A14.1.09.321 |
| FMA | 275020 |
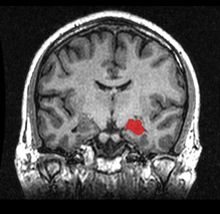
The hippocampus (named after its resemblance to the seahorse, from the Greek ἱππόκαμπος, "seahorse" from ἵππος hippos, "horse" and κάμπος kampos, "sea monster") is a major component of the brains of humans and other vertebrates. Humans and other mammals have two hippocampi, one in each side of the brain. The hippocampus belongs to the limbic system and plays important roles in the consolidation of information from short-term memory to long-term memory, and in spatial navigation. The hippocampus is located under the cerebral cortex (sub-cortical)[1] and in primates in the medial temporal lobe. It contains two main interlocking parts: the hippocampus proper (also called Ammon's horn)[2] and the dentate gyrus.
In Alzheimer's disease, the hippocampus is one of the first regions of the brain to suffer damage; memory loss and disorientation are included among the early symptoms. Damage to the hippocampus can also result from oxygen starvation (hypoxia), encephalitis, or medial temporal lobe epilepsy. People with extensive, bilateral hippocampal damage may experience anterograde amnesia—the inability to form and retain new memories.
In rodents as model organisms, the hippocampus has been studied extensively as part of a brain system responsible for spatial memory and navigation. Many neurons in the rat and mouse hippocampus respond as place cells: that is, they fire bursts of action potentials when the animal passes through a specific part of its environment. Hippocampal place cells interact extensively with head direction cells, whose activity acts as an inertial compass, and conjecturally with grid cells in the neighboring entorhinal cortex.
Since different neuronal cell types are neatly organized into layers in the hippocampus, it has frequently been used as a model system for studying neurophysiology. The form of neural plasticity known as long-term potentiation (LTP) was first discovered to occur in the hippocampus and has often been studied in this structure. LTP is widely believed to be one of the main neural mechanisms by which memory is stored in the brain.
Name
The earliest description of the ridge running along the floor of the temporal horn of the lateral ventricle comes from the Venetian anatomist Julius Caesar Aranzi (1587), who likened it first to a silkworm and then to a seahorse (Latin: hippocampus from Greek: ἵππος, "horse" and κάμπος, "sea monster"). The German anatomist Duvernoy (1729), the first to illustrate the structure, also wavered between "seahorse" and "silkworm." "Ram's horn" was proposed by the Danish anatomist Jacob Winsløw in 1732; and a decade later his fellow Parisian, the surgeon de Garengeot, used "cornu Ammonis" – horn of (the ancient Egyptian god) Amun.[4]
Another reference appeared with the term pes hippocampi, which may date back to Diemerbroeck in 1672, introducing a comparison with the shape of the folded back forelimbs and webbed feet of the mythological hippocampus (Greek: ἱππόκαμπος), a sea monster with a horse's forequarters and a fish's tail. The hippocampus was then described as pes hippocampi major, with an adjacent bulge in the occipital horn, the calcar avis, being named pes hippocampi minor.[4] The renaming of the hippocampus as hippocampus major, and the calcar avis as hippocampus minor, has been attributed to Félix Vicq-d'Azyr systematising nomenclature of parts of the brain in 1786. Mayer mistakenly used the term hippopotamus in 1779, and was followed by some other authors until Karl Friedrich Burdach resolved this error in 1829. In 1861 the hippocampus minor became the centre of a dispute over human evolution between Thomas Henry Huxley and Richard Owen, satirised as the Great Hippocampus Question. The term hippocampus minor fell from use in anatomy textbooks, and was officially removed in the Nomina Anatomica of 1895.[5]
Today, the structure is called the hippocampus rather than hippocampus major, with pes hippocampi often being regarded as synonymous with De Garengeot's "cornu Ammonis",[4] a term that survives in the names of the four main histological divisions of the hippocampus: CA1, CA2, CA3, and CA4.[6]
Anatomy
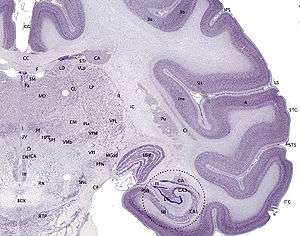
In terms of anatomy, the hippocampus is an elaboration of the edge of the cerebral cortex.[7] The set of structures that line the edge of the cortex make up the limbic system (Latin limbus = border): These include the hippocampus, cingulate cortex, olfactory cortex, and amygdala. Paul MacLean once suggested, as part of his triune brain theory, that the limbic structures comprise the neural basis of emotion. Some neuroscientists no longer believe that the concept of a unified "limbic system" is valid, however.[8] Yet, the hippocampus is anatomically connected to parts of the brain that are involved with emotional behavior—the septum, the hypothalamic mammillary body, and the anterior nuclear complex in the thalamus—therefore its role as a limbic structure cannot be completely dismissed.
The hippocampus as a whole has the shape of a curved tube, which has been variously compared to a seahorse, a ram's horn (Cornu Ammonis, hence the subdivisions CA1 through CA4), or a banana.[7] It can be distinguished as a zone where the cortex narrows into a single layer of densely packed pyramidal neurons 3 to 6 cells deep in rats, which curl into a tight U shape; one edge of the "U," field CA4, is embedded into a backward-facing, strongly flexed, V-shaped cortex, the dentate gyrus. It consists of ventral and dorsal portions, both of which are of similar composition but are parts of different neural circuits.[9] This general layout holds across the full range of mammalian species, from hedgehog to human, although the details vary. In the rat, the two hippocampi resemble a pair of bananas, joined at the stems by the hippocampal commissure that crosses the midline under the anterior corpus callosum. In human or monkey brains, the portion of the hippocampus down at the bottom, near the base of the temporal lobe, is much broader than the part at the top. One of the consequences of this complex geometry is that cross-sections through the hippocampus can show a variety of shapes, depending on the angle and location of the cut.
.jpg)
.png)
The entorhinal cortex (EC), located in the parahippocampal gyrus, is considered to be part of the hippocampal region because of its anatomical connections. The EC is strongly and reciprocally connected with many other parts of the cerebral cortex. In addition, the medial septal nucleus, the anterior nuclear complex and nucleus reuniens of the thalamus and the supramammillary nucleus of the hypothalamus, as well as the raphe nuclei and locus coeruleus in the brainstem send axons to the EC. The main output pathway (perforant path, first described by Ramon y Cajal) of EC axons comes from the large pyramidal cells in layer II that "perforate" the subiculum and project densely to the granule cells in the dentate gyrus, apical dendrites of CA3 get a less dense projection, and the apical dendrites of CA1 get a sparse projection. Thus, the perforant path establishes the EC as the main "interface" between the hippocampus and other parts of the cerebral cortex. The dentate granule cell axons (called mossy fibers) pass on the information from the EC on thorny spines that exit from the proximal apical dendrite of CA3 pyramidal cells. Then, CA3 axons (the Schaffer collaterals) exit from the deep part of the cell body and loop up into the region where the apical dendrites are located, then extend to CA1. CA1 then sends axons back to the EC - the only region to do so - completing the trisynaptic circuit. Within the hippocampus, the flow of information from the EC is largely unidirectional, with signals propagating through a series of tightly packed cell layers, first to the dentate gyrus, then to the CA3 layer, then to the CA1 layer, then to the subiculum, then out of the hippocampus to the EC, mainly due to collateralization of the CA3 axons. Each of these layers also contains complex intrinsic circuitry and extensive longitudinal connections.[7]
Several other connections play important roles in hippocampal function.[7] Beyond the output to the EC, additional output pathways go to other cortical areas including the prefrontal cortex. A very important large output goes to the lateral septal area and to the mammillary body of the hypothalamus. The hippocampus receives modulatory input from the serotonin, norepinephrine, and dopamine systems, and from the nucleus reuniens of the thalamus to field CA1. A very important projection comes from the medial septal area, which sends cholinergic and GABAergic fibers to all parts of the hippocampus. The inputs from the septal area play a key role in controlling the physiological state of the hippocampus; destruction of the septal area abolishes the hippocampal theta rhythm and severely impairs certain types of memory.[10]
The cortical region adjacent to the hippocampus is known collectively as the parahippocampal gyrus (or parahippocampus).[11] It includes the EC and also the perirhinal cortex, which derives its name from the fact that it lies next to the rhinal sulcus. The perirhinal cortex plays an important role in visual recognition of complex objects. There is also substantial evidence that it makes a contribution to memory, which can be distinguished from the contribution of the hippocampus. It is apparent that complete amnesia occurs only when both the hippocampus and the parahippocampus are damaged.[11]
Functions
Historically, the earliest widely held hypothesis was that the hippocampus is involved in olfaction.[12] This idea was cast into doubt by a series of anatomical studies that did not find any direct projections to the hippocampus from the olfactory bulb.[13] However, later work did confirm that the olfactory bulb does project into the ventral part of the lateral entorhinal cortex, and field CA1 in the ventral hippocampus sends axons to the main olfactory bulb,[14] the anterior olfactory nucleus, and to the primary olfactory cortex. There continues to be some interest in hippocampal olfactory responses, in particular the role of the hippocampus in memory for odors, but few specialists today believe that olfaction is its primary function.[15][16]
Over the years, three main ideas of hippocampal function have dominated the literature: inhibition, memory, and space. The behavioral inhibition theory (caricatured by John O'Keefe and Lynn Nadel as "slam on the brakes!")[17] was very popular up to the 1960s. It derived much of its justification from two observations: first, that animals with hippocampal damage tend to be hyperactive; second, that animals with hippocampal damage often have difficulty learning to inhibit responses that they have previously been taught, especially if the response requires remaining quiet as in a passive avoidance test. Jeffrey Gray developed this line of thought into a full-fledged theory of the role of the hippocampus in anxiety.[18] The inhibition theory is currently the least popular of the three.[19]
The second major line of thought relates the hippocampus to memory. Although it had historical precursors, this idea derived its main impetus from a famous report by William Beecher Scoville and Brenda Milner[20] describing the results of surgical destruction of the hippocampi (in an attempt to relieve epileptic seizures), in Henry Molaison,[21] known until his death in 2008 as "Patient H.M." The unexpected outcome of the surgery was severe anterograde and partial retrograde amnesia; Molaison was unable to form new episodic memories after his surgery and could not remember any events that occurred just before his surgery, but he did retain memories of events that occurred many years earlier extending back into his childhood. This case attracted such widespread professional interest that Molaison became the most intensively studied subject in medical history.[22] In the ensuing years, other patients with similar levels of hippocampal damage and amnesia (caused by accident or disease) have also been studied, and thousands of experiments have studied the physiology of activity-driven changes in synaptic connections in the hippocampus. There is now universal agreement that the hippocampi play some sort of important role in memory; however, the precise nature of this role remains widely debated.[23][24]
The third important theory of hippocampal function relates the hippocampus to space. The spatial theory was originally championed by O'Keefe and Nadel, who were influenced by E.C. Tolman's theories about "cognitive maps" in humans and animals. O'Keefe and his student Dostrovsky in 1971 discovered neurons in the rat hippocampus that appeared to them to show activity related to the rat's location within its environment.[25] Despite skepticism from other investigators, O'Keefe and his co-workers, especially Lynn Nadel, continued to investigate this question, in a line of work that eventually led to their very influential 1978 book The Hippocampus as a Cognitive Map.[26] There is now almost universal agreement that hippocampal function plays an important role in spatial coding, but the details are widely debated.[27]
Role in memory
Psychologists and neuroscientists generally agree that the hippocampus plays an important role in the formation of new memories about experienced events (episodic or autobiographical memory).[24][28] Part of this function is hippocampal involvement in the detection of novel events, places and stimuli.[29] Some researchers regard the hippocampus as part of a larger medial temporal lobe memory system responsible for general declarative memory (memories that can be explicitly verbalized—these would include, for example, memory for facts in addition to episodic memory).[23]
Due to bilateral symmetry the brain has a hippocampus in each cerebral hemisphere. If damage to the hippocampus occurs in only one hemisphere, leaving the structure intact in the other hemisphere, the brain can retain near-normal memory functioning.[30] Severe damage to the hippocampi in both hemispheres results in profound difficulties in forming new memories (anterograde amnesia) and often also affects memories formed before the damage occurred (retrograde amnesia). Although the retrograde effect normally extends many years back before the brain damage, in some cases older memories remain. This retention of older memories leads to the idea that consolidation over time involves the transfer of memories out of the hippocampus to other parts of the brain.[31]
Damage to the hippocampus does not affect some types of memory, such as the ability to learn new skills (playing a musical instrument or solving certain types of puzzles, for example). This fact suggests that such abilities depend on different types of memory (procedural memory) and different brain regions. Furthermore, amnesic patients frequently show "implicit" memory for experiences even in the absence of conscious knowledge. For example, patients asked to guess which of two faces they have seen most recently may give the correct answer most of the time in spite of stating that they have never seen either of the faces before. Some researchers distinguish between conscious recollection, which depends on the hippocampus, and familiarity, which depends on portions of the medial temporal lobe.[32]
Role in spatial memory and navigation

Studies conducted on freely moving rats and mice have shown that many hippocampal neurons have "place fields", that is, they fire bursts of action potentials when a rat passes through a particular part of the environment. Evidence for place cells in primates is limited, perhaps in part because it is difficult to record brain activity from freely moving monkeys. Place-related hippocampal neural activity has been reported in monkeys moving around inside a room while seated in a restraint chair;[33] on the other hand, Edmund Rolls and his colleagues instead described hippocampal cells that fire in relation to the place a monkey is looking at, rather than the place where its body is located.[34] In humans, cells with location-specific firing patterns have been reported in a study of patients with drug-resistant epilepsy who were undergoing an invasive procedure to localize the source of their seizures, with a view to surgical resection. The patients had diagnostic electrodes implanted in their hippocampus and then used a computer to move around in a virtual reality town.[35]
Place responses in rats and mice have been studied in hundreds of experiments over four decades, yielding a large quantity of information.[27] Place cell responses are shown by pyramidal cells in the hippocampus proper, and granule cells in the dentate gyrus. These constitute the great majority of neurons in the densely packed hippocampal layers. Inhibitory interneurons, which make up most of the remaining cell population, frequently show significant place-related variations in firing rate that are much weaker than those displayed by pyramidal or granule cells. There is little if any spatial topography in the representation; in general, cells lying next to each other in the hippocampus have uncorrelated spatial firing patterns. Place cells are typically almost silent when a rat is moving around outside the place field but reach sustained rates as high as 40 Hertz when the rat is near the center. Neural activity sampled from 30 to 40 randomly chosen place cells carries enough information to allow a rat's location to be reconstructed with high confidence. The size of place fields varies in a gradient along the length of the hippocampus, with cells at the dorsal end showing the smallest fields, cells near the center showing larger fields, and cells at the ventral tip fields that cover the entire environment.[27] In some cases, the firing rate of rat hippocampal cells depends not only on place but also on the direction a rat is moving, the destination toward which it is traveling, or other task-related variables.[36]
The discovery of place cells in the 1970s led to a theory that the hippocampus might act as a cognitive map—a neural representation of the layout of the environment.[37] Several lines of evidence support the hypothesis. It is a frequent observation that without a fully functional hippocampus, humans may not remember where they have been and how to get where they are going: Getting lost is one of the most common symptoms of amnesia.[38] Studies with animals have shown that an intact hippocampus is required for initial learning and long-term retention of some spatial memory tasks, in particular ones that require finding the way to a hidden goal.[39][40][41][42] The "cognitive map hypothesis" has been further advanced by recent discoveries of head direction cells, grid cells, and border cells in several parts of the rodent brain that are strongly connected to the hippocampus.[27][43]
Brain imaging shows that people have more active hippocampi when correctly navigating, as tested in a computer-simulated "virtual" navigation task.[44] Also, there is evidence that the hippocampus plays a role in finding shortcuts and new routes between familiar places. For example, London's taxi drivers must learn a large number of places and the most direct routes between them (they have to pass a strict test, The Knowledge, before being licensed to drive the famous black cabs). A study at University College London by Maguire, et al.. (2000)[45] showed that part of the hippocampus is larger in taxi drivers than in the general public, and that more experienced drivers have bigger hippocampi. Whether having a bigger hippocampus helps an individual to become a better cab driver, or if finding shortcuts for a living makes an individual's hippocampus grow is yet to be elucidated. However, in that study, Maguire et al. examined the correlation between size of the grey matter and length of time that had been spent as a taxi driver, and found a positive correlation between the length of time an individual had spent as a taxi driver and the volume of the right hippocampus. It was found that the total volume of the hippocampus remained constant, from the control group vs. taxi drivers. That is to say that the posterior portion of a taxi driver's hippocampus is indeed increased, but at the expense of the anterior portion. There have been no known detrimental effects reported from this disparity in hippocampal proportions.[45]
Hippocampal formation
Various sections of the hippocampal formation are shown to be functionally and anatomically distinct. The dorsal (DH), ventral (VH) and intermediate regions of the hippocampal formation serve different functions, project with differing pathways, and have varying degrees of place field neurons.[46] The dorsal region of the hippocampal formation serves for spatial memory, verbal memory, and learning of conceptual information. Using the radial arm maze, Pothuizen et al. (2004) found lesions in the DH to cause spatial memory impairment while VH lesions did not. Its projecting pathways include the medial septal complex and supramammillary nucleus.[47] The dorsal hippocampal formation also has more place field neurons than both the ventral and intermediate hippocampal formations.[48]
The intermediate hippocampus has overlapping characteristics with both the ventral and dorsal hippocampus.[46] Using anterograde tracing methods, Cenquizca and Swanson (2007) located the moderate projections to two primary olfactory cortical areas and prelimbic areas of the mPFC. This region has the smallest number of place field neurons. The ventral hippocampus functions in fear conditioning and affective processes.[49] Anagnostaras et al. (2002) showed that alterations to the ventral hippocampus reduced the amount of information sent to the amygdala by the dorsal and ventral hippocampus, consequently altering fear conditioning in rats.[50]
Physiology
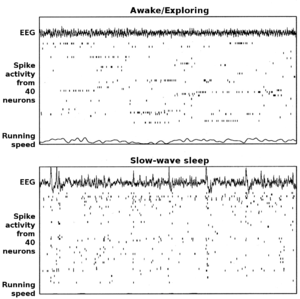
The hippocampus shows two major "modes" of activity, each associated with a distinct pattern of neural population activity and waves of electrical activity as measured by an electroencephalogram (EEG). These modes are named after the EEG patterns associated with them: theta and large irregular activity (LIA). The main characteristics described below are for the rat, which is the animal most extensively studied.[51]
The theta mode appears during states of active, alert behavior (especially locomotion), and also during REM (dreaming) sleep.[52] In the theta mode, the EEG is dominated by large regular waves with a frequency range of 6 to 9 Hertz, and the main groups of hippocampal neurons (pyramidal cells and granule cells) show sparse population activity, which means that in any short time interval, the great majority of cells are silent, while the small remaining fraction fire at relatively high rates, up to 50 spikes in one second for the most active of them. An active cell typically stays active for half a second to a few seconds. As the rat behaves, the active cells fall silent and new cells become active, but the overall percentage of active cells remains more or less constant. In many situations, cell activity is determined largely by the spatial location of the animal, but other behavioral variables also clearly influence it.
The LIA mode appears during slow-wave (non-dreaming) sleep, and also during states of waking immobility such as resting or eating.[52] In the LIA mode, the EEG is dominated by sharp waves that are randomly timed large deflections of the EEG signal lasting for 25–50 milliseconds. Sharp waves are frequently generated in sets, with sets containing up to 5 or more individual sharp waves and lasting up to 500 ms. The spiking activity of neurons within the hippocampus is highly correlated with sharp wave activity. Most neurons decrease their firing rate between sharp waves; however, during a sharp wave, there is a dramatic increase of firing rate in up to 10% of the hippocampal population
These two hippocampal activity modes can be seen in primates as well as rats, with the exception that it has been difficult to see robust theta rhythmicity in the primate hippocampus. There are, however, qualitatively similar sharp waves and similar state-dependent changes in neural population activity.[53]
Theta rhythm
Because of its densely packed neural layers, the hippocampus generates some of the largest EEG signals as theta waves of any brain structure, which generate the hippocampal theta rhythm.[54] In some situations the EEG is dominated by regular waves at 3 to 10 Hertz, often continuing for many seconds. These reflect subthreshold membrane potentials and strongly modulate the spiking of hippocampal neurons and synchronise across the hippocampus in a travelling wave pattern.[55] The trisynaptic circuit is a relay of neurotransmission in the hippocampus that interacts with many brain regions. From rodent studies it has been proposed that the trisynaptic circuit generates the hippocampal theta rhythm.[56]
Theta rhythmicity is very obvious in rabbits and rodents and also clearly present in cats and dogs. Whether theta can be seen in primates is not yet clear.[57] In rats (the animals that have been the most extensively studied), theta is seen mainly in two conditions: first, when an animal is walking or in some other way actively interacting with its surroundings; second, during REM sleep.[58] The function of theta has not yet been convincingly explained although numerous theories have been proposed.[51] The most popular hypothesis has been to relate it to learning and memory. An example would be the phase with which theta rhythms, at the time of stimulation of a neuron, shape the effect of that stimulation upon its synapses. What is meant here is that theta rhythms may affect those aspects of learning and memory that are dependent upon synaptic plasticity.[59] It is well established that lesions of the medial septum—the central node of the theta system—cause severe disruptions of memory. However, the medial septum is more than just the controller of theta; it is also the main source of cholinergic projections to the hippocampus.[7] It has not been established that septal lesions exert their effects specifically by eliminating the theta rhythm.[60]
Sharp waves
During sleep or during waking states when an animal is resting or otherwise not engaged with its surroundings, the hippocampal EEG shows a pattern of irregular slow waves, somewhat larger in amplitude than theta waves. This pattern is occasionally interrupted by large surges called sharp waves.[61] These events are associated with bursts of spike activity lasting 50 to 100 milliseconds in pyramidal cells of CA3 and CA1. They are also associated with short-lived high-frequency EEG oscillations called "ripples", with frequencies in the range 150 to 200 Hertz in rats. Sharp waves are most frequent during sleep when they occur at an average rate of around 1 per second (in rats) but in a very irregular temporal pattern. Sharp waves are less frequent during inactive waking states and are usually smaller. Sharp waves have also been observed in humans and monkeys. In macaques, sharp waves are robust but do not occur as frequently as in rats.[53]
One of the most interesting aspects of sharp waves is that they appear to be associated with memory. Wilson and McNaughton 1994,[62] and numerous later studies, reported that when hippocampal place cells have overlapping spatial firing fields (and therefore often fire in near-simultaneity), they tend to show correlated activity during sleep following the behavioral session. This enhancement of correlation, commonly known as reactivation, has been found to occur mainly during sharp waves.[63] It has been proposed that sharp waves are, in fact, reactivations of neural activity patterns that were memorized during behavior, driven by strengthening of synaptic connections within the hippocampus.[64] This idea forms a key component of the "two-stage memory" theory, advocated by Buzsáki and others, which proposes that memories are stored within the hippocampus during behavior and then later transferred to the neocortex during sleep. Sharp waves are suggested to drive Hebbian synaptic changes in the neocortical targets of hippocampal output pathways.[65] Suppression of sharp wave/ripple complexes in sleep or during immobility can interfere with memories expressed at the level of the behavior,[66][67] nonetheless, the newly formed CA1 place cell code can re-emerge even after a sleep with abolished sharp-wave/ripples in spatially non-demanding tasks.[68]
Long-term potentiation
Since at least the time of Ramon y Cajal, psychologists have speculated that the brain stores memory by altering the strength of connections between neurons that are simultaneously active.[69] This idea was formalized by Donald Hebb in 1948,[70] but for many years thereafter, attempts to find a brain mechanism for such changes failed. In 1973, Tim Bliss and Terje Lømo described a phenomenon in the rabbit hippocampus that appeared to meet Hebb's specifications: a change in synaptic responsiveness induced by brief strong activation and lasting for hours or days or longer.[71] This phenomenon was soon referred to as long-term potentiation, abbreviated LTP. As a candidate mechanism for memory, LTP has since been studied intensively, and a great deal has been learned about it.
The hippocampus is a particularly favorable site for studying LTP because of its densely packed and sharply defined layers of neurons, but similar types of activity-dependent synaptic change have now been observed in many other brain areas.[72] The best-studied form of LTP occurs at synapses that terminate on dendritic spines and use the neurotransmitter glutamate. Several of the major pathways within the hippocampus fit this description and exhibit LTP.[73] The synaptic changes depend on a special type of glutamate receptor, the NMDA receptor, which has the special property of allowing calcium to enter the postsynaptic spine only when presynaptic activation and postsynaptic depolarization occur at the same time.[74] Drugs that interfere with NMDA receptors block LTP and have major effects on some types of memory, especially spatial memory. Transgenic mice, genetically modified in ways that disable the LTP mechanism, also generally show severe memory deficits.[74]
Pathology
Aging
Age-related conditions such as Alzheimer's disease (for which hippocampal disruption is one of the earliest signs[75]) have a severe impact on many types of cognition, but even normal aging is associated with a gradual decline in some types of memory, including episodic memory and working memory (or short-term memory). Because the hippocampus is thought to play a central role in memory, there has been considerable interest in the possibility that age-related declines could be caused by hippocampal deterioration.[76] Some early studies reported substantial loss of neurons in the hippocampus of elderly people, but later studies using more precise techniques found only minimal differences.[76] Similarly, some MRI studies have reported shrinkage of the hippocampus in elderly people, but other studies have failed to reproduce this finding. There is, however, a reliable relationship between the size of the hippocampus and memory performance — meaning that not all elderly people show hippocampal shrinkage, but those who do tend to perform less well on some memory tasks.[77] There are also reports that memory tasks tend to produce less hippocampal activation in elderly than in young subjects.[77] Furthermore, a randomized-control study published in 2011 found that aerobic exercise could increase the size of the hippocampus in adults aged 55 to 80 and also improve spatial memory.[78]
Stress
The hippocampus contains high levels of glucocorticoid receptors, which make it more vulnerable to long-term stress than most other brain areas.[79] Stress-related steroids affect the hippocampus in at least three ways: first, by reducing the excitability of some hippocampal neurons; second, by inhibiting the genesis of new neurons in the dentate gyrus; third, by causing atrophy of dendrites in pyramidal cells of the CA3 region. There is evidence that humans having experienced severe, long-lasting traumatic stress show atrophy of the hippocampus more than of other parts of the brain.[80] These effects show up in post-traumatic stress disorder,[81] and they may contribute to the hippocampal atrophy reported in schizophrenia[82] and severe depression.[83] A recent study has also revealed atrophy as a result of depression, but this can be stopped with anti-depressants even if they are not effective in relieving other symptoms.[84] Hippocampal atrophy is also frequently seen in Cushing's syndrome, a disorder caused by high levels of cortisol in the bloodstream. At least some of these effects appear to be reversible if the stress is discontinued. There is, however, evidence derived mainly from studies using rats that stress occurring shortly after birth can affect hippocampal function in ways that persist throughout life.[85]
Sex-specific responses to stress have also been demonstrated to have an effect on the hippocampus. During situations in which adult male and female rats were exposed to chronic stress the females were shown to be better able to cope.[86]
Epilepsy
The hippocampus is often the focus of epileptic seizures: hippocampal sclerosis is the most commonly visible type of tissue damage in temporal lobe epilepsy.[87] It is not yet clear, however, whether the epilepsy is usually caused by hippocampal abnormalities or whether the hippocampus is damaged by cumulative effects of seizures.[88] In experimental settings where repetitive seizures are artificially induced in animals, hippocampal damage is a frequent result. This may be a consequence of the hippocampus's being one of the most electrically excitable parts of the brain. It may also have something to do with the fact that the hippocampus is one of very few brain regions where new neurons continue to be created throughout life.[89]
Schizophrenia
The causes of schizophrenia are not at all well understood, but numerous abnormalities of brain structure have been reported. The most thoroughly investigated alterations involve the cerebral cortex, but effects on the hippocampus have also been described. Many reports have found reductions in the size of the hippocampus in schizophrenic subjects.[90] The changes probably result from altered development rather than tissue damage and show up even in subjects never having been medicated. Several lines of evidence implicate changes in synaptic organization and connectivity.[90] It is unclear whether hippocampal alterations play any role in causing the psychotic symptoms that are the most important feature of schizophrenia. Anthony Grace and his co-workers have suggested, on the basis of experimental work using animals, that hippocampal dysfunction might produce an alteration of dopamine release in the basal ganglia, thereby indirectly affecting the integration of information in the prefrontal cortex.[91] Others have suggested that hippocampal dysfunction might account for disturbances in long-term memory frequently observed in people with schizophrenia.[92]
Transient global amnesia
A current hypothesis as to one cause of transient global amnesia—a dramatic, sudden, temporary, near-total loss of short-term memory—is that it may be due to venous congestion of the brain,[93] leading to ischemia of structures such as the hippocampus that are involved in memory.[94]
Evolution
The hippocampus has a generally similar appearance across the range of mammal species, from monotremes such as the echidna to primates such as humans.[95] The hippocampal-size-to-body-size ratio broadly increases, being about twice as large for primates as for the echidna. It does not, however, increase at anywhere close to the rate of the neocortex-to-body-size ratio. Therefore, the hippocampus takes up a much larger fraction of the cortical mantle in rodents than in primates. In adult humans the volume of the hippocampus on each side of the brain is about 3.0 to 3.5 cm3 as compared to 320 to 420 cm3 for the volume of the neocortex.[96]
There is also a general relationship between the size of the hippocampus and spatial memory. When comparisons are made between similar species, those that have a greater capacity for spatial memory tend to have larger hippocampal volumes.[97] This relationship also extends to sex differences; in species where males and females show strong differences in spatial memory ability they also tend to show corresponding differences in hippocampal volume.[98]
Non-mammalian species do not have a brain structure that looks like the mammalian hippocampus, but they have one that is considered homologous to it. The hippocampus, as pointed out above, is in essence the medial edge of the cortex. Only mammals have a fully developed cortex, but the structure it evolved from, called the pallium, is present in all vertebrates, even the most primitive ones such as the lamprey or hagfish.[99] The pallium is usually divided into three zones: medial, lateral and dorsal. The medial pallium forms the precursor of the hippocampus. It does not resemble the hippocampus visually because the layers are not warped into an S shape or enfolded by the dentate gyrus, but the homology is indicated by strong chemical and functional affinities. There is now evidence that these hippocampal-like structures are involved in spatial cognition in birds, reptiles, and fish.[100]
In birds, the correspondence is sufficiently well established that most anatomists refer to the medial pallial zone as the "avian hippocampus".[101] Numerous species of birds have strong spatial skills, in particular those that cache food. There is evidence that food-caching birds have a larger hippocampus than other types of birds and that damage to the hippocampus causes impairments in spatial memory.[102]
The story for fish is more complex. In teleost fish (which make up the great majority of existing species), the forebrain is distorted in comparison to other types of vertebrates: Most neuroanatomists believe that the teleost forebrain is in essence everted, like a sock turned inside-out, so that structures that lie in the interior, next to the ventricles, for most vertebrates, are found on the outside in teleost fish, and vice versa.[103] One of the consequences of this is that the medial pallium ("hippocampal" zone) of a typical vertebrate is thought to correspond to the lateral pallium of a typical fish. Several types of fish (particularly goldfish) have been shown experimentally to have strong spatial memory abilities, even forming "cognitive maps" of the areas they inhabit.[97] There is evidence that damage to the lateral pallium impairs spatial memory.[104][105]
Thus, the role of the hippocampal region in navigation appears to begin far back in vertebrate evolution, predating splits that occurred hundreds of millions of years ago.[106] It is not yet known whether the medial pallium plays a similar role in even more primitive vertebrates, such as sharks and rays, or even lampreys and hagfish. Some types of insects, and molluscs such as the octopus, also have strong spatial learning and navigation abilities, but these appear to work differently from the mammalian spatial system, so there is as yet no good reason to think that they have a common evolutionary origin; nor is there sufficient similarity in brain structure to enable anything resembling a "hippocampus" to be identified in these species. Some have proposed, however, that the insect's mushroom bodies may have a function similar to that of the hippocampus.[107]
Notes
- ↑ Wright, Anthony. Chapter 5: Limbic System: Hippocampus. Department of Neurobiology and Anatomy, The UT Medical School at Houston
- ↑ Pearce, 2001
- ↑ preparation by László Seress in 1980.
- 1 2 3 Duvernoy, 2005
- ↑ Gross, 1993
- ↑ Wechsler, 2004
- 1 2 3 4 5 Amaral and Lavenex, 2006
- ↑ Kötter & Stephan, 1997
- ↑ Moser and Moser, 1998
- ↑ Winson, 1978
- 1 2 Eichenbaum et al, 2007
- ↑ Finger, S (2001). "Defining and controlling the circuits of emotion". Origins of neuroscience: a history of explorations into brain function. Oxford/NewYork: Oxford University Press. p. 286. ISBN 0-19-506503-4.
- ↑ Finger, p. 183
- ↑ "Extrinsic projections from area CA1 of the rat hippocampus: olfactory, cortical, subcortical, and bilateral hippocampal formation projections". Journal of Comparative Neurology. 1990. doi:10.1002/cne.903020308.
- ↑ Eichenbaum et al, 1991
- ↑ Vanderwolf, 2001
- ↑ Nadel et al., 1975
- ↑ Gray and McNaughton, 2000
- ↑ Best & White, 1999
- ↑ Scoville and Milner, 1957
- ↑ New York Times, 12-06-2008
- ↑ Squire, 2009
- 1 2 Squire, 1992
- 1 2 Eichenbaum and Cohen, 1993
- ↑ O'Keefe and Dostrovsky, 1971
- ↑ O'Keefe and Nadel, 1978
- 1 2 3 4 Moser et al., 2008
- ↑ Squire and Schacter, 2002
- ↑ VanElzakker et al., 2008
- ↑ Di Gennaro G, Grammaldo LG, Quarato PP, Esposito V, Mascia A, Sparano A, Meldolesi GN, Picardi A (Jun 2006). "Severe amnesia following bilateral medial temporal lobe damage occurring on two distinct occasions". Neurological Sciences. 27 (2): 129–33. doi:10.1007/s10072-006-0614-y. PMID 16816912.
- ↑ Squire and Schacter, 2002, Ch. 1
- ↑ Diana et al., 2007
- ↑ Matsumara et al., 1999
- ↑ Rolls and Xiang, 2006
- ↑ Ekstrom et al., 2003
- ↑ Smith and Mizumori, 2006
- ↑ O'Keefe and Nadel
- ↑ Chiu et al., 2004
- ↑ Morris et al., 1982
- ↑ Sutherland et al., 1982
- ↑ Sutherland et al., 2001
- ↑ Clark et al., 2005
- ↑ Solstad et al., 2008
- ↑ Maguire et al., 1998
- 1 2 Maguire et al., 2000
- 1 2 Fanselow, 2010
- ↑ Pothuizen et al., 2004
- ↑ Jung et al., 1994
- ↑ Cenquizca et al., 2007
- ↑ Anagnostaras et al., 2002
- 1 2 Buzsáki, 2006
- 1 2 Buzsáki et al., 1990
- 1 2 Skaggs et al., 2007
- ↑ Buzsáki, 2002
- ↑ Lubenov & Siapas, 2009
- ↑ Komisaruk, B. R. (1970). "Synchrony between limbic system theta activity and rhythmical behavior in rats". Journal of comparative and physiological psychology. 70 (3): 482.
- ↑ Cantero et al., 2003
- ↑ Vanderwolf, 1969
- ↑ Huerta & Lisman, 1993
- ↑ Kahana et al., 2001
- ↑ Buzsáki, 1986
- ↑ Wilson & McNaughton, 1994
- ↑ Jackson et al., 2006
- ↑ Sutherland & McNaughton, 2000
- ↑ Buzsáki, 1989
- ↑ Girardeau, Gabrielle; Karim Benchenane; Sidney I Wiener; György Buzsáki; Michaël B Zugaro (September 2009). "Selective suppression of hippocampal ripples impairs spatial memory". Nature Neuroscience. 12 (10): 1222–1223. doi:10.1038/nn.2384. PMID 19749750.
- ↑ Ego-Stengel, Valérie; Matthew A. Wilson (January 2010). "Disruption of ripple-associated hippocampal activity during rest impairs spatial learning in the rat". Hippocampus. 20 (1): 1–10. doi:10.1002/hipo.20707. PMC 2801761
 . PMID 19816984.
. PMID 19816984. - ↑ Kovacs KA, O'Neill J, Schoenenberger P, Penttonen M, Ranguel Guerrero DK, Csicsvari J (19 Nov 2016). "Optogenetically Blocking Sharp Wave Ripple Events in Sleep Does Not Interfere with the Formation of Stable Spatial Representation in the CA1 Area of the Hippocampus". PLoS One. doi:10.1371/journal.pone.0164675. PMID 27760158.
- ↑ Ramon y Cajal, 1894
- ↑ Hebb, 1948
- ↑ Bliss & Lømo, 1973
- ↑ Cooke & Bliss, 2006
- ↑ Malenka & Bear, 2004
- 1 2 Nakazawa et al., 2004
- ↑ Hampel et al., 2008
- 1 2 Prull et al., 2000, p. 105
- 1 2 Prull et al., 2000, p. 107
- ↑ Erickson et al., 2011
- ↑ Joels, 2008
- ↑ Fu et al, 2010
- ↑ Karl A, Schaefer M, Malta LS, Dörfel D, Rohleder N, Werner A (2006). "A meta-analysis of structural brain abnormalities in PTSD". Neuroscience and Biobehavioral Reviews. 30 (7): 1004–31. doi:10.1016/j.neubiorev.2006.03.004. PMID 16730374.
- ↑ Wright IC, Rabe-Hesketh S, Woodruff PW, David AS, Murray RM, Bullmore ET (January 2000). "Meta-analysis of regional brain volumes in schizophrenia". The American Journal of Psychiatry. 157 (1): 16–25. doi:10.1176/ajp.157.1.16. PMID 10618008.
- ↑ Kempton MJ, Salvador Z, Munafò MR, Geddes JR, Simmons A, Frangou S, Williams SC (Jul 2011). "Structural neuroimaging studies in major depressive disorder. Meta-analysis and comparison with bipolar disorder". Archives of General Psychiatry. 68 (7): 675–90. doi:10.1001/archgenpsychiatry.2011.60. PMID 21727252. see also MRI database at www.depressiondatabase.org
- ↑ Campbell & MacQueen, 2004
- ↑ Garcia-Segura, pp. 170–71
- ↑ Conrad CD (2008). "Chronic stress-induced hippocampal vulnerability: the glucocorticoid vulnerability hypothesis". Reviews in the Neurosciences. 19 (6): 395–411. doi:10.1515/revneuro.2008.19.6.395. PMC 2746750
 . PMID 19317179.
. PMID 19317179. - ↑ Chang and Lowenstein, 2003
- ↑ Sloviter, 2005
- ↑ Kuruba et al., 2009
- 1 2 Harrison, 2004
- ↑ Goto & Grace, 2008
- ↑ Boyer et al., 2007
- ↑ Lewis SL (Aug 1998). "Aetiology of transient global amnesia". Lancet. 352 (9125): 397–9. doi:10.1016/S0140-6736(98)01442-1. PMID 9717945.
- ↑ Chung CP, Hsu HY, Chao AC, Chang FC, Sheng WY, Hu HH (Jun 2006). "Detection of intracranial venous reflux in patients of transient global amnesia". Neurology. 66 (12): 1873–77. doi:10.1212/01.wnl.0000219620.69618.9d. PMID 16801653.
- ↑ West, 1990
- ↑ Suzuki et al, 2005
- 1 2 Jacobs, 2003
- ↑ Jacobs et al., 1990
- ↑ Aboitiz et al., 2003
- ↑ Rodríguez et al., 2002
- ↑ Colombo and Broadbent, 2000
- ↑ Shettleworth, 2003
- ↑ Nieuwenhuys, 1982
- ↑ Portavella et al., 2002
- ↑ Vargas et al., 2006
- ↑ Broglio et al., 2005
- ↑ Mizunami et al., 1998
References
- Aboitiz F, Morales D, Montiel J (Oct 2003). "The evolutionary origin of the mammalian isocortex: towards an integrated developmental and functional approach". The Behavioral and Brain Sciences. 26 (5): 535–52. doi:10.1017/S0140525X03000128. PMID 15179935.
- Amaral D, Lavenex P (2006). "Ch 3. Hippocampal Neuroanatomy". In Andersen P, Morris R, Amaral D, Bliss T, O'Keefe J. The Hippocampus Book. Oxford University Press. ISBN 978-0-19-510027-3.
- Anagnostaras SG, Gale GD, Fanselow MS (2002). "The hippocampus and Pavlovian fear conditioning: reply to Bast et al" (PDF). Hippocampus. 12 (4): 561–565. doi:10.1002/hipo.10071. PMID 12201641.
- Best PJ, White AM (1999). "Placing hippocampal single-unit studies in a historical context". Hippocampus. 9 (4): 346–51. doi:10.1002/(SICI)1098-1063(1999)9:4<346::AID-HIPO2>3.0.CO;2-3. PMID 10495017.
- Bliss TV, Lomo T (Jul 1973). "Long-lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path". The Journal of Physiology. 232 (2): 331–56. doi:10.1113/jphysiol.1973.sp010273. PMC 1350458
 . PMID 4727084.
. PMID 4727084. - Boyer P, Phillips JL, Rousseau FL, Ilivitsky S (Apr 2007). "Hippocampal abnormalities and memory deficits: new evidence of a strong pathophysiological link in schizophrenia". Brain Research Reviews. 54 (1): 92–112. doi:10.1016/j.brainresrev.2006.12.008. PMID 17306884.
- Broglio C, Gómez A, Durán E, Ocaña FM, Jiménez-Moya F, Rodríguez F, Salas C (Sep 2005). "Hallmarks of a common forebrain vertebrate plan: specialized pallial areas for spatial, temporal and emotional memory in actinopterygian fish". Brain Research Bulletin. 66 (4-6): 397–99. doi:10.1016/j.brainresbull.2005.03.021. PMID 16144602.
- Burke SN, Barnes CA (Jan 2006). "Neural plasticity in the ageing brain". Nature Reviews. Neuroscience. 7 (1): 30–40. doi:10.1038/nrn1809. PMID 16371948.
- Buzsáki G (Nov 1986). "Hippocampal sharp waves: their origin and significance". Brain Research. 398 (2): 242–52. doi:10.1016/0006-8993(86)91483-6. PMID 3026567.
- Buzsáki G (1989). "Two-stage model of memory trace formation: a role for "noisy" brain states". Neuroscience. 31 (3): 551–70. doi:10.1016/0306-4522(89)90423-5. PMID 2687720.
- Buzsáki G, Chen LS, Gage FH (1990). "Spatial organization of physiological activity in the hippocampal region: relevance to memory formation". Progress in Brain Research. Progress in Brain Research. 83: 257–68. doi:10.1016/S0079-6123(08)61255-8. ISBN 9780444811493. PMID 2203100.
- Buzsáki G (Jan 2002). "Theta oscillations in the hippocampus" (PDF). Neuron. 33 (3): 325–40. doi:10.1016/S0896-6273(02)00586-X. PMID 11832222.
- Buzsáki G (2006). Rhythms of the Brain. Oxford University Press. ISBN 0-19-530106-4.
- Ramón y Cajal S (1894). "The Croonian Lecture: La Fine Structure des Centres Nerveux". Proceedings of the Royal Society. 55 (331–335): 444–68. doi:10.1098/rspl.1894.0063.
- Campbell S, Macqueen G (Nov 2004). "The role of the hippocampus in the pathophysiology of major depression". Journal of Psychiatry & Neuroscience. 29 (6): 417–26. PMC 524959
 . PMID 15644983.
. PMID 15644983. - Cantero JL, Atienza M, Stickgold R, Kahana MJ, Madsen JR, Kocsis B (Nov 2003). "Sleep-dependent theta oscillations in the human hippocampus and neocortex". The Journal of Neuroscience. 23 (34): 10897–903. PMID 14645485.
- Carey B (2008-12-04). "H. M., an Unforgettable Amnesiac, Dies at 82". The New York Times. Retrieved 2009-04-27.
- Chiu YC, Algase D, Whall A, Liang J, Liu HC, Lin KN, Wang PN (2004). "Getting lost: directed attention and executive functions in early Alzheimer's disease patients". Dementia and Geriatric Cognitive Disorders. 17 (3): 174–80. doi:10.1159/000076353. PMID 14739541.
- Chang BS, Lowenstein DH (Sep 2003). "Epilepsy". The New England Journal of Medicine. 349 (13): 1257–66. doi:10.1056/NEJMra022308. PMID 14507951.
- Cho RY, Gilbert A, Lewis DA (2005). "Ch 22. The neurobiology of schizophrenia". In Charney DS, Nestler EJ. Neurobiology of Mental Illness. Oxford University Press US. ISBN 978-0-19-518980-3.
- Cenquizca LA, Swanson LW (Nov 2007). "Spatial organization of direct hippocampal field CA1 axonal projections to the rest of the cerebral cortex". Brain Research Reviews. 56 (1): 1–26. doi:10.1016/j.brainresrev.2007.05.002. PMC 2171036
 . PMID 17559940.
. PMID 17559940. - Clark RE, Broadbent NJ, Squire LR (2005). "Hippocampus and remote spatial memory in rats". Hippocampus. 15 (2): 260–72. doi:10.1002/hipo.20056. PMC 2754168
 . PMID 15523608.
. PMID 15523608. - Colombo M, Broadbent N (Jun 2000). "Is the avian hippocampus a functional homologue of the mammalian hippocampus?". Neuroscience and Biobehavioral Reviews. 24 (4): 465–84. doi:10.1016/S0149-7634(00)00016-6. PMID 10817844.
- Cooke SF, Bliss TV (Jul 2006). "Plasticity in the human central nervous system". Brain. 129 (Pt 7): 1659–73. doi:10.1093/brain/awl082. PMID 16672292.
- de Olmos J, Hardy H, Heimer L (Sep 1978). "The afferent connections of the main and the accessory olfactory bulb formations in the rat: an experimental HRP-study". The Journal of Comparative Neurology. 181 (2): 213–244. doi:10.1002/cne.901810202. PMID 690266.
- Diana RA, Yonelinas AP, Ranganath C (Sep 2007). "Imaging recollection and familiarity in the medial temporal lobe: a three-component model". Trends in Cognitive Sciences. 11 (9): 379–86. doi:10.1016/j.tics.2007.08.001. PMID 17707683.
- Duvernoy HM (2005). "Introduction". The Human Hippocampus (3rd ed.). Berlin: Springer-Verlag. p. 1. ISBN 3-540-23191-9.
- Eichenbaum H, Otto TA, Wible CG, Piper JM (1991). "Ch 7. Building a model of the hippocampus in olfaction and memory". In Davis JL, Eichenbaum H,. Olfaction. MIT Press. ISBN 978-0-262-04124-9.
- Eichenbaum H, Cohen NJ (1993). Memory, Amnesia, and the Hippocampal System. MIT Press.
- Eichenbaum H, Yonelinas AP, Ranganath C (2007). "The medial temporal lobe and recognition memory". Annual Review of Neuroscience. 30: 123–52. doi:10.1146/annurev.neuro.30.051606.094328. PMC 2064941
 . PMID 17417939.
. PMID 17417939. - Ekstrom AD, Kahana MJ, Caplan JB, Fields TA, Isham EA, Newman EL, Fried I (Sep 2003). "Cellular networks underlying human spatial navigation" (PDF). Nature. 425 (6954): 184–88. Bibcode:2003Natur.425..184E. doi:10.1038/nature01964. PMID 12968182.
- Erickson KI, et al. (Feb 2011). "Exercise training increases size of hippocampus and improves memory". Proceedings of the National Academy of Sciences of the United States of America. 108 (7): 3017–3022. Bibcode:2011PNAS..108.3017E. doi:10.1073/pnas.1015950108. PMC 3041121
 . PMID 21282661.
. PMID 21282661. - Fanselow MS, Dong HW (Jan 2010). "Are the dorsal and ventral hippocampus functionally distinct structures?" (PDF). Neuron. 65 (1): 7–19. doi:10.1016/j.neuron.2009.11.031. PMC 2822727
 . PMID 20152109.
. PMID 20152109. - Finger, S (2001). Origins of Neuroscience: A History of Explorations Into Brain Function. Oxford University Press US. ISBN 978-0-19-514694-3.
- Garcia-Segura LM (2009). Hormones and Brain Plasticity. Oxford University Press US. ISBN 978-0-19-532661-1.
- Woon FL, Sood S, Hedges DW (Oct 2010). "Hippocampal volume deficits associated with exposure to psychological trauma and posttraumatic stress disorder in adults: a meta-analysis". Progress in Neuro-Psychopharmacology & Biological Psychiatry. 34 (7): 1181–1188. doi:10.1016/j.pnpbp.2010.06.016. PMID 20600466.
- Gorwood P, Corruble E, Falissard B, Goodwin GM (Jun 2008). "Toxic effects of depression on brain function: impairment of delayed recall and the cumulative length of depressive disorder in a large sample of depressed outpatients". The American Journal of Psychiatry. 165 (6): 731–9. doi:10.1176/appi.ajp.2008.07040574. PMID 18381906.
- Goto Y, Grace AA (Nov 2008). "Limbic and cortical information processing in the nucleus accumbens". Trends in Neurosciences. 31 (11): 552–8. doi:10.1016/j.tins.2008.08.002. PMC 2884964
 . PMID 18786735.
. PMID 18786735. - Gray, JA; McNaughton N (2000). The Neuropsychology of Anxiety: An Enquiry into the Functions of the Septo-Hippocampal System. Oxford University Press.
- Gross CG (Oct 1993). "Hippocampus minor and man's place in nature: a case study in the social construction of neuroanatomy". Hippocampus. 3 (4): 403–416. doi:10.1002/hipo.450030403. PMID 8269033.
- Hampel H, Bürger K, Teipel SJ, Bokde AL, Zetterberg H, Blennow K (Jan 2008). "Core candidate neurochemical and imaging biomarkers of Alzheimer's disease". Alzheimer's & Dementia. 4 (1): 38–48. doi:10.1016/j.jalz.2007.08.006. PMID 18631949.
- Harrison PJ (Jun 2004). "The hippocampus in schizophrenia: a review of the neuropathological evidence and its pathophysiological implications". Psychopharmacology. 174 (1): 151–62. doi:10.1007/s00213-003-1761-y. PMID 15205886.
- Hebb DO (1949). Organization of Behavior: a Neuropsychological Theory. New York: John Wiley. ISBN 0-471-36727-3.
- Huerta PT, Lisman JE (Aug 1993). "Heightened synaptic plasticity of hippocampal CA1 neurons during a cholinergically induced rhythmic state". Nature. 364 (6439): 723–5. Bibcode:1993Natur.364..723H. doi:10.1038/364723a0. PMID 8355787.
- Jackson JC, Johnson A, Redish AD (Nov 2006). "Hippocampal sharp waves and reactivation during awake states depend on repeated sequential experience". The Journal of Neuroscience. 26 (48): 12415–26. doi:10.1523/JNEUROSCI.4118-06.2006. PMID 17135403.
- Jacobs LF, Gaulin SJ, Sherry DF, Hoffman GE (Aug 1990). "Evolution of spatial cognition: sex-specific patterns of spatial behavior predict hippocampal size". Proceedings of the National Academy of Sciences of the United States of America. 87 (16): 6349–52. Bibcode:1990PNAS...87.6349J. doi:10.1073/pnas.87.16.6349. PMC 54531
 . PMID 2201026.
. PMID 2201026. - Jacobs LF (2003). "The evolution of the cognitive map". Brain, Behavior and Evolution. 62 (2): 128–39. doi:10.1159/000072443. PMID 12937351.
- Jung MW, Wiener SI, McNaughton BL (Dec 1994). "Comparison of spatial firing characteristics of units in dorsal and ventral hippocampus of the rat" (PDF). The Journal of Neuroscience. 14 (12): 7347–7356. PMID 7996180.
- Kahana MJ, Seelig D, Madsen JR (Dec 2001). "Theta returns". Current Opinion in Neurobiology. 11 (6): 739–44. doi:10.1016/S0959-4388(01)00278-1. PMID 11741027.
- Kötter R, Stephan KE (1997). "Useless or helpful? The "limbic system" concept". Reviews in the Neurosciences. 8 (2): 139–45. doi:10.1515/REVNEURO.1997.8.2.139. PMID 9344183.
- Joëls M (Apr 2008). "Functional actions of corticosteroids in the hippocampus". European Journal of Pharmacology. 583 (2-3): 312–321. doi:10.1016/j.ejphar.2007.11.064. PMID 18275953.
- Kuruba R, Hattiangady B, Shetty AK (Jan 2009). "Hippocampal neurogenesis and neural stem cells in temporal lobe epilepsy". Epilepsy & Behavior. 14 Suppl 1: 65–73. doi:10.1016/j.yebeh.2008.08.020. PMC 2654382
 . PMID 18796338.
. PMID 18796338. - Lubenov EV, Siapas AG (May 2009). "Hippocampal theta oscillations are travelling waves". Nature. 459 (7246): 534–9. doi:10.1038/nature08010. PMID 19489117.
- Maguire EA, Burgess N, Donnett JG, Frackowiak RS, Frith CD, O'Keefe J (May 1998). "Knowing where and getting there: a human navigation network". Science. 280 (5365): 921–24. Bibcode:1998Sci...280..921M. doi:10.1126/science.280.5365.921. PMID 9572740.
- Maguire EA, Gadian DG, Johnsrude IS, Good CD, Ashburner J, Frackowiak RS, Frith CD (Apr 2000). "Navigation-related structural change in the hippocampi of taxi drivers". Proceedings of the National Academy of Sciences of the United States of America. 97 (8): 4398–403. Bibcode:2000PNAS...97.4398M. doi:10.1073/pnas.070039597. PMC 18253
 . PMID 10716738.
. PMID 10716738. - Malenka RC, Bear MF (Sep 2004). "LTP and LTD: an embarrassment of riches". Neuron. 44 (1): 5–21. doi:10.1016/j.neuron.2004.09.012. PMID 15450156.
- Matsumura N, Nishijo H, Tamura R, Eifuku S, Endo S, Ono T (Mar 1999). "Spatial- and task-dependent neuronal responses during real and virtual translocation in the monkey hippocampal formation". The Journal of Neuroscience. 19 (6): 2381–93. PMID 10066288.
- McNaughton BL, Battaglia FP, Jensen O, Moser EI, Moser MB (Aug 2006). "Path integration and the neural basis of the 'cognitive map'". Nature Reviews. Neuroscience. 7 (8): 663–78. doi:10.1038/nrn1932. PMID 16858394.
- Mizunami M, Weibrecht JM, Strausfeld NJ (Dec 1998). "Mushroom bodies of the cockroach: their participation in place memory". The Journal of Comparative Neurology. 402 (4): 520–37. doi:10.1002/(SICI)1096-9861(19981228)402:4<520::AID-CNE6>3.0.CO;2-K. PMID 9862324.
- Morris RG, Garrud P, Rawlins JN, O'Keefe J (Jun 1982). "Place navigation impaired in rats with hippocampal lesions". Nature. 297 (5868): 681–83. Bibcode:1982Natur.297..681M. doi:10.1038/297681a0. PMID 7088155.
- Moser MB, Moser EI (1998). "Functional differentiation in the hippocampus". Hippocampus. 8 (6): 608–19. doi:10.1002/(SICI)1098-1063(1998)8:6<608::AID-HIPO3>3.0.CO;2-7. PMID 9882018.
- Moser EI, Kropff E, Moser MB (2008). "Place cells, grid cells, and the brain's spatial representation system". Annual Review of Neuroscience. 31: 69. doi:10.1146/annurev.neuro.31.061307.090723. PMID 18284371.
- Nadel L, O'Keefe J, Black A (Jun 1975). "Slam on the brakes: a critique of Altman, Brunner, and Bayer's response-inhibition model of hippocampal function". Behavioral Biology. 14 (2): 151–62. doi:10.1016/S0091-6773(75)90148-0. PMID 1137539.
- Nakazawa K, McHugh TJ, Wilson MA, Tonegawa S (May 2004). "NMDA receptors, place cells and hippocampal spatial memory". Nature Reviews. Neuroscience. 5 (5): 361–72. doi:10.1038/nrn1385. PMID 15100719.
- Nieuwenhuys, R (1982). "An Overview of the Organization of the Brain of Actinopterygian Fishes". Am. Zool. 22 (2): 287–310. doi:10.1093/icb/22.2.287.
- O'Kane G, Kensinger EA, Corkin S (2004). "Evidence for semantic learning in profound amnesia: an investigation with patient H.M". Hippocampus. 14 (4): 417–25. doi:10.1002/hipo.20005. PMID 15224979.
- O'Keefe J, Dostrovsky J (Nov 1971). "The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat". Brain Research. 34 (1): 171–75. doi:10.1016/0006-8993(71)90358-1. PMID 5124915.
- O'Keefe, J; Nadel L (1978). The Hippocampus as a Cognitive Map. Oxford University Press.
- Portavella M, Vargas JP, Torres B, Salas C (2002). "The effects of telencephalic pallial lesions on spatial, temporal, and emotional learning in goldfish". Brain Research Bulletin. 57 (3-4): 397–99. doi:10.1016/S0361-9230(01)00699-2. PMID 11922997.
- Pearce JM (Sep 2001). "Ammon's horn and the hippocampus". Journal of Neurology, Neurosurgery, and Psychiatry. 71 (3): 351. doi:10.1136/jnnp.71.3.351. PMC 1737533
 . PMID 11511709.
. PMID 11511709. - Pothuizen HH, Zhang WN, Jongen-Rêlo AL, Feldon J, Yee BK (Feb 2004). "Dissociation of function between the dorsal and the ventral hippocampus in spatial learning abilities of the rat: a within-subject, within-task comparison of reference and working spatial memory". The European Journal of Neuroscience. 19 (3): 705–712. doi:10.1111/j.0953-816X.2004.03170.x. PMID 14984421.
- Prull MW, Gabrieli JD, Bunge SA (2000). "Ch 2. Age-related changes in memory: A cognitive neuroscience perspective". In Craik FI, Salthouse TA. The handbook of aging and cognition. Erlbaum. ISBN 978-0-8058-2966-2.
- Rodríguez F, López JC, Vargas JP, Broglio C, Gómez Y, Salas C (2002). "Spatial memory and hippocampal pallium through vertebrate evolution: insights from reptiles and teleost fish". Brain Research Bulletin. 57 (3-4): 499–503. doi:10.1016/S0361-9230(01)00682-7. PMID 11923018.
- Rolls ET, Xiang JZ (2006). "Spatial view cells in the primate hippocampus and memory recall". Reviews in the Neurosciences. 17 (1-2): 175–200. doi:10.1515/REVNEURO.2006.17.1-2.175. PMID 16703951.
- Rosenzweig ES, Barnes CA (Feb 2003). "Impact of aging on hippocampal function: plasticity, network dynamics, and cognition". Progress in Neurobiology. 69 (3): 143–79. doi:10.1016/S0301-0082(02)00126-0. PMID 12758108.
- Scoville WB, Milner B (Feb 1957). "Loss of recent memory after bilateral hippocampal lesions". Journal of Neurology, Neurosurgery, and Psychiatry. 20 (1): 11–21. doi:10.1136/jnnp.20.1.11. PMC 497229
 . PMID 13406589.
. PMID 13406589. - Shettleworth SJ (2003). "Memory and hippocampal specialization in food-storing birds: challenges for research on comparative cognition". Brain, Behavior and Evolution. 62 (2): 108–16. doi:10.1159/000072441. PMID 12937349.
- Skaggs WE, McNaughton BL, Wilson MA, Barnes CA (1996). "Theta phase precession in hippocampal neuronal populations and the compression of temporal sequences". Hippocampus. 6 (2): 149–76. doi:10.1002/(SICI)1098-1063(1996)6:2<149::AID-HIPO6>3.0.CO;2-K. PMID 8797016.
- Skaggs WE, McNaughton BL, Permenter M, Archibeque M, Vogt J, Amaral DG, Barnes CA (Aug 2007). "EEG sharp waves and sparse ensemble unit activity in the macaque hippocampus". Journal of Neurophysiology. 98 (2): 898–910. doi:10.1152/jn.00401.2007. PMID 17522177.
- Sloviter RS (Feb 2005). "The neurobiology of temporal lobe epilepsy: too much information, not enough knowledge". Comptes Rendus Biologies. 328 (2): 143–53. doi:10.1016/j.crvi.2004.10.010. PMID 15771000.
- Smith DM, Mizumori SJ (2006). "Hippocampal place cells, context, and episodic memory". Hippocampus. 16 (9): 716–29. doi:10.1002/hipo.20208. PMID 16897724.
- Solstad T, Boccara CN, Kropff E, Moser MB, Moser EI (Dec 2008). "Representation of geometric borders in the entorhinal cortex". Science. 322 (5909): 1865–68. Bibcode:2008Sci...322.1865S. doi:10.1126/science.1166466. PMID 19095945.
- Squire LR (Apr 1992). "Memory and the hippocampus: a synthesis from findings with rats, monkeys, and humans". Psychological Review. 99 (2): 195–231. doi:10.1037/0033-295X.99.2.195. PMID 1594723.
- Squire, LR; Schacter DL (2002). The Neuropsychology of Memory. Guilford Press.
- Squire LR (Jan 2009). "The legacy of patient H.M. for neuroscience". Neuron. 61 (1): 6–9. doi:10.1016/j.neuron.2008.12.023. PMC 2649674
 . PMID 19146808.
. PMID 19146808. - Sutherland GR, McNaughton B (Apr 2000). "Memory trace reactivation in hippocampal and neocortical neuronal ensembles". Current Opinion in Neurobiology. 10 (2): 180–86. doi:10.1016/S0959-4388(00)00079-9. PMID 10753801.
- Sutherland RJ, Kolb B, Whishaw IQ (Aug 1982). "Spatial mapping: definitive disruption by hippocampal or medial frontal cortical damage in the rat". Neuroscience Letters. 31 (3): 271–6. doi:10.1016/0304-3940(82)90032-5. PMID 7133562.
- Sutherland RJ, Weisend MP, Mumby D, Astur RS, Hanlon FM, Koerner A, Thomas MJ, Wu Y, Moses SN, Cole C, Hamilton DA, Hoesing JM (2001). "Retrograde amnesia after hippocampal damage: recent vs. remote memories in two tasks". Hippocampus. 11 (1): 27–42. doi:10.1002/1098-1063(2001)11:1<27::AID-HIPO1017>3.0.CO;2-4. PMID 11261770.
- Suzuki M, Hagino H, Nohara S, Zhou SY, Kawasaki Y, Takahashi T, Matsui M, Seto H, Ono T, Kurachi M (Feb 2005). "Male-specific volume expansion of the human hippocampus during adolescence". Cerebral Cortex. 15 (2): 187–93. doi:10.1093/cercor/bhh121. PMID 15238436.
- Vanderwolf CH (Dec 2001). "The hippocampus as an olfacto-motor mechanism: were the classical anatomists right after all?". Behavioural Brain Research. 127 (1-2): 25–47. doi:10.1016/S0166-4328(01)00354-0. PMID 11718883.
- Vargas JP, Bingman VP, Portavella M, López JC (Nov 2006). "Telencephalon and geometric space in goldfish". The European Journal of Neuroscience. 24 (10): 2870–78. doi:10.1111/j.1460-9568.2006.05174.x. PMID 17156211.
- VanElzakker M, Fevurly RD, Breindel T, Spencer RL (Dec 2008). "Environmental novelty is associated with a selective increase in Fos expression in the output elements of the hippocampal formation and the perirhinal cortex". Learning & Memory. 15 (12): 899–908. doi:10.1101/lm.1196508. PMC 2632843
 . PMID 19050162.
. PMID 19050162. - Wechsler RT, Morss, AM, Wustoff, CJ, & Caughey, AB (2004). Blueprints notes & cases: Neuroscience. Oxford: Blackwell Publishing. p. 37. ISBN 1-4051-0349-3.
- West MJ (1990). "Stereological studies of the hippocampus: a comparison of the hippocampal subdivisions of diverse species including hedgehogs, laboratory rodents, wild mice and men". Progress in Brain Research. Progress in Brain Research. 83: 13–36. doi:10.1016/S0079-6123(08)61238-8. ISBN 9780444811493. PMID 2203095.
- Wilson MA, McNaughton BL (Jul 1994). "Reactivation of hippocampal ensemble memories during sleep". Science. 265 (5172): 676–79. Bibcode:1994Sci...265..676W. doi:10.1126/science.8036517. PMID 8036517.
- Winson J (Jul 1978). "Loss of hippocampal theta rhythm results in spatial memory deficit in the rat". Science. 201 (4351): 160–63. Bibcode:1978Sci...201..160W. doi:10.1126/science.663646. PMID 663646.
Further reading
Journals
- Hippocampus (Wiley)
Books
- Per Andersen; Richard Morris; David Amaral; Tim Bliss; John O'Keefe, eds. (2007). The Hippocampus Book. Oxford University Press. ISBN 978-0-19-510027-3.
- Dori Derdikman; James J. Knierim, eds. (2014). Space,Time and Memory in the Hippocampal Formation. Springer. ISBN 978-3-7091-1292-2.
- Henri M. Duvernoy; F. Cattin (2005). The Human Hippocampus: Functional Anatomy, Vascularization, and Serial Sections with MRI. Springer. ISBN 978-3-540-23191-2.
- Howard Eichenbaum (2002). The Cognitive Neuroscience of Memory. Oxford University Press US. ISBN 978-0-19-514175-7.
- edited by Patricia E. Sharp. (2002). Patricia E. Sharp, ed. The Neural Basis of Navigation: Evidence from Single Cell Recording. Springer. ISBN 978-0-7923-7579-1.
- Philippe Taupin (2007). The Hippocampus: Neurotransmission and Plasticity in the Nervous System. Nova Publishers. ISBN 978-1-60021-914-6.
- John H Byrne, ed. (2008). Learning and Memory: A comprehensive reference. Elsevier. ISBN 978-0-12-370509-9.
External links
| Wikimedia Commons has media related to Hippocampus. |
- Stained brain slice images which include the "hippocampus" at the BrainMaps project
- Diagram of a Hippocampal Brain Slice
- Hippocampus – Cell Centered Database
- Temporal-lobe.com An interactive diagram of the rat parahippocampal-hippocampal region
- Search Hippocampus on BrainNavigator via BrainNavigator
- Gyorgy Buzsaki (2010) Hippocampus. Scholarpedia. 6(1):1468.